NCERT Solutions For Class 11 Chemistry Chapter 13 Hydrocarbons Long Question And Answers
Question 1. How can an eclipsed conformation of ethane be converted into a staggered conformation?
Answer:
In an ethane molecule, if one carbon atom is kept fixed around the C—C bond axis and the other carbon atom is rotated at a minimum angle of 60°, then the eclipsed conformation is converted to the staggered conformation.

Question 2. Give examples of a chiral conformation and an achiral conformation of n-butane.
Answer:
Gauche-staggered conformation of n-butane is chiral because it cannot be superimposed on its mirror image.
However, the fully eclipsed conformation of n-butane is achiral as it can be superimposed on its mirror image.

Question 3. Arrange the following conformations of n-butane according to their increasing stability:
- Gauche-staggered
- Fully eclipsed
- Eclipsed and
- Ante-Mggered
Answer:
When 2-iodopropane is used as the alkyl halide in the Wurtz reaction, the alkane obtained is 2,3-dimethylbutane.

Question 4. Which of the following alkanes cannot be prepared by the Wurtz reaction in good yield?
- (CH3)2CHCH2CH(CH3)2
- (CH3)2CHCH2CH2CH(CH3)2
- (CH3)3CCH2CH2CH2CH3
- CH3CH2C(CH3)2CH2CH3
- (CH3)3C-C(CH3)3
Answer:
(1), (3) and (4) are three unsymmetrical alkanes. So, these cannot be prepared by Wurtz reaction in good yield. Again, for preparing alkane (5), a 3° alkyl halide is required. So, despite being a symmetrical alkane, (5) cannot be prepared by the Wurtz reaction.
NCERT Class 11 Chemistry Chapter 13 Hydrocarbons Long Question and Answers
Question 5. How will you prepare methane and ethane starting from ethanoic acid?
Answer:

Question 6. How many monochloro derivatives are obtained on chlorination of n-pentane, isopentane and neopentane? Write down their structures.
Answer:
There are three and four types of non-equivalent hydrogen atoms in n-pentane (CH3CH2CH2CH2CH3) and isopentane [CH3CH(CH3)CH2CH3] respectively. Whereas, in neopentane [(CH3)4C], all H -atoms are equivalent.
Therefore, chlorination of n-pentane, isopentane and neopentane form three, four and one monochloride derivatives respectively.

Question 7. Chlorination of cyclohexane to prepare chlorocyclohexane is more practicable than the chlorination of methylcyclohexane to prepare l-chloro-l-methylcyclohexane— explain.
Answer:
There are five types of non-equivalent H -atoms in methylcyclohexane
![]()
When it undergoes chlorination, four other monochloride derivatives are formed along with 1-chloro-1-methylcyclohexane

As a result, the yield of the desired product is low’—’ and Low it is difficult to separate the product from the mixture. On the other hand, all H -atoms in cyclohexane are equivalent and thus, only chlorocyclohexane
![]()
Is formed as the product for this reason, Achlorination of cyclohexane to prepare chlorocyclohexane is more feasible than the chlorination of methylcyclohexane to prepare 1-chloro-l-methylcyclohexane.
Question 8. Write the IUPAC names of the following compounds:

Answer:

Question 9. Write the structural formula:
- 3-(1- methyl ethyl) hex-2-ene;
- 4-ethyl- 2, 4- dimethyl kept-1- ene
Answer:

Question 10. Write the IUPAC names and structures of the alkenes having the molecular formula C5H10.
Answer:
IUPAC names and structures of the alkenes having the molecular formula C5H10 are as follows

Hydrocarbons Class 11 Long Question and Answers NCERT
Question 11. Write the mechanism of acid-catalysed dehydration of butyl alcohol.
Answer:
Dehydration of isopropyl alcohol in the presence of concentrated H2SO4 is an El reaction. The reaction occurs in three steps. The second step of the reaction is the slowest, i.e., it is the die rate-determining step of the reaction.
Step 1: Protonation of the alcohol.

Step 2: Elimination of water molecules and formation of carbocation

Step 3: Elimination of proton from carbocation

Question 12. Write the structures of A and B obtained from given reactions
Answer:

Answer:
A is R —CHBr —CH3 and B is RCHBrCH2Br. The alkene, HBr and the formed alkyl bromide (A) are all colourless. So, the left reaction cannot be used to detect ethylenic unsaturation.
On the other hand, the alkene and the formed dibromoalkane (B) are colourless but bromine has a reddish-brown colour.
So, the right reaction can be used to detect ethylenic unsaturation because decolourisation of bromine takes place in this reaction.

Question 13. How can a double bond be created in a molecule of a compound which has a carbon-carbon single bond?
Answer:
A double bond is created in a molecule of a compound containing a carbon-carbon single bond by the can-given method.

Question 14. Which reaction is used to detect ethylenic unsaturation and why? Write the structures and IUPAC names of the compounds expected to be obtained in the given reactions:
⇒ \(\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}_2+\mathrm{HCl}→{\text { Peroxide }}\)
⇒ \(\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}_2+\mathrm{HBr}→{\text { Peroxide }}\)
Answer:

Question 15. Write the product of the given reaction. Explain its formation:

Answer:

Due to the -R and -I -effect of the — NO2 group, carbocation [I] is less stable than carbocation [II]. So the reaction proceeds through carbocation [II] and the major product formed is

Question 16. State Markownikoffs rule. Explain with an example. How would you convert ethylene to acetylene? Identify the compound in the reaction—

Answer:
Markownikoffs rule and example: Conversion of ethylene to acetylene

Question 17. Ethane can be dried by passing through concentrated H2S04 but not ethylene—why?
Answer:
Ethane being a saturated hydrocarbon does not react with concentrated H2SO4
CH3 —CH3 (Ethane) + cone. H2SO4 . So, ethane can be dried by passing through concentrated H2SO4.
On the other hand, ethylene being an unsaturated hydrocarbon, when passed through concentrated H2SO4 gets absorbed by the acid and forms ethyl hydrogen sulphate. So, ethylene cannot be dried by passing through concentrated H2SO4.

Question 18. Identify the compounds A, B and C in the following reaction and write their names:

Answer:
Carbonyl compounds B and C contain three and two carbon atoms respectively. There are also three carbon atoms on one side of the double bond and two carbon atoms on the other side of the double bond in the alkene.
Therefore, two alkenes with molecular formula C5H10 are: 2-methylbut-2-ene [CH3 —C(CH3)=CHCH3] and pent-2-ene (CH3CH2CH=CHCH3) . If A (C5H10) is 2-methylbut-2-ene, thenB (C3H6O) &C(C2H4O) areacetone (CH3COCH3) and acetaldehyde (CH3CHO) respectively.

If A is pent-2-ene, then B and C are propanal (CH3CH2CHO) and acetaldehyde (CH3CHO) respectively.

Question 19. An alkene on ozonolysis produces propanone and propanal. The alkene is—
- 2-methyl pent-2-ene
- 3-methyl pent-2- ene
- A-methyl pent-2-ene
- Hex-3-ene.
Answer:
The products formed on ozonolysis are propanone and propanal. Therefore, the alkene can be determined as—

The alkene is (1) 2-methylpent-2-ene.
Question 20. How can cis- and trans-hydroxylation of cis-2-butene be carried out? Comment on the optical activity of the formed products.
Answer:
Osmium tetroxide adds to the double bond of cis-2- butene to form osmic ester which is hydrolysed by an aqueous ethanolic solution of sodium bisulphite. In this case, the two —OH groups get attached to the doubly bonded carbon atoms from the same side of the double bond and form 1,2-diol.
So, cis-hydroxylation takes place in case of this reaction. On the other hand, cis-2-butene reacts with peracids to form the corresponding epoxide. The resulting epoxide on hydrolysis with dilute acid or alkali yields 1,2-diol.
Epoxidation followed by hydrolysis causes the addition of two —OH groups from the opposite sides of the double bond. So, trans-hydroxylation takes place in case of this reaction.

Question 21. What product would you expect from the following reaction?

Answer:
An alkyne should be formed when a vicinal dihalide is refluxed with ethanolic KOH. However, in the given case, an alkyne does not form because a six-membered ring system cannot accommodate a linear portion like C—C=C —C. So, the compound formed is 1,3-cyclohexadiene.

Question 22. Identify and B in the following reactions

Answer:
Question 23. Give IUPAC names of the following compounds:
1. CH3CH2-CH=C=CH2
2.

3. CH2=CH-CH(CH3)-(CH3)C=CH2
Answer:
IUPACname: 1,2-pentadiene.
IUPAC name: 1-methyl-1,4-cyclohexadiene.
IUPAC name: 2,3-dimethyl-1,4-pentadiene
Question 24. What are the two planar conformations of 1,3-butadiene? Which conformation is less stable and why?
Answer:
The two planar conformations of1,3-butadiene are—

Due to steric hindrance or strain, s-ds-conformation is less stable.
Question 25. Between 1,3- and 1,4-cyclohexadiene, which compound has a lower value of heat of hydrogenation and why?
Answer:
1,3-cyclohexadiene being a conjugated diene is more stable than 1,4-cyclohexadiene which is a nonconjugated diene. So, the heat of hydrogenation of 1,3- cyclohexadiene has a lesser value than that of 1,4- cyclohexadiene.

Question 26. Calculate the double bond equivalent of benzene from its molecular formula.
Answer:
Double bond equivalent (DBE) of compound,
⇒ \(\mathrm{DBE}=\frac{\sum n(v-2)}{2}+1\)
Where n is the number of different atoms present in the molecule and v is the valency of each atom. The molecular formula of benzene is C6H6.
So, DBE of benzene = \(\frac{6(4-2)+6(1-2)}{2}+1=4\)
Class 11 Chemistry Chapter 13 Hydrocarbons NCERT Long Q&A
Question 27. What knowledge about the carbon-carbon bond length in benzene may be obtained from valence bond theory?
Answer:

The benzene molecule is a resonance hybrid of Kekule structures (1) and (2) and the contribution of each hybrid structure is 50% i.e., equal.
The single bonds (C—C) and the double bonds (C—C) in structure (1) become double and single bonds respectively in structure (2). As the two equally stable resonance structures (1) and (2) contribute equally to the hybrid.
It may be said that any two adjacent carbon atoms of a benzene molecule are linked by a bond intermediate between a single and a double bond. So, all the carbon-carbon bonds of benzene are equivalent and their lengths are equal (1.39A).
Again, the bond order of each bond is the same (1.5). So, it can be said that all carbon-carbon bonds are equal in length.
⇒ \(\text { Bond order }=\frac{\text { Double bond }+ \text { Single bond }}{2}=\frac{2+1}{2}=1.5\)
Question 28. Which of the following representations is correct and why?

Answer:
According to representation
(1), it seems that structures (1) and (2) have a separate existence. There is no separate existence of structures (1) and (2).
So, (1) and (2) cannot be related by ’. Thus, the representation (i) is incorrect. Since (1) and (2) are two resonance structures which have no separate existence. So, (1) and (2) can be related by’ •*-»ÿ ‘. Thus, the representation
(2) is correct.
Question 29. What is the basic difference between aromatic and anti¬ aromatic compounds?
Answer:
Monocyclic planar conjugated polyene systems containing (4n + 2) delocalised; π-electrons (n = 0, 1,2,3, are called aromatic compounds.

Monocyclic planar conjugated polyene systems containing 4n delocalised π- electrons (n = 1,2,3,…) are called antiaromatic compounds.

Question 30. Will cyclooctatetraene exhibit aromatic character? Explain.
Answer:
Since cyclooctatetraene does not contain (4n + 2)n electrons, it does not exhibit an aromatic character.
As cyclooctatetraene has 4n – electrons (n=2), it should be an antiaromatic compound. However, the ring of this compound is very large so it does not exist in the unstable planar shape, rather it forms a tub-shaped structure. As a result, conjugation is lost and so cyclooctatetraene is a non-aromatic compound.

Question 31. Using the theory of aromaticity, explain the finding that A and B are different compounds, but Cand D are identical.
Answer:
As A is an antiaromatic compound (4nπ -electron system, n = 1 ), it becomes unstable due to the delocalisation of π electrons. As delocalisation of π -electrons does not take place for A, B is not the resonance structure of A.
B is the structural isomer of A. So, A and B are two different compounds. Again, C is an aromatic compound [(4n + 2)π electron-system, n = 1 ] which attains stability due to the delocalisation of electrons. So, delocalisation of electrons takes place for C. D is the resonance structure of C, i.e., Cand D are same compound.

Question 32. Classify each of the given species as aromatic, antiaromatic and nonaromatic

Answer:
- Is an antiaromatic compound because the B atom contains a vacant p -orbital,
- Is a non-aromatic compound because one carbon atom of the ring does not have a p -orbital.
- Behaves as an aromatic compound with (4n + 2)π-electrons (n = 1) because of the vacant d -d-orbital and lone pair of electrons of the sulphur atom

Is an aromatic ion with (4n+2)π- electrons, n = 0
Question 33. Which is the smallest aromatics species?
Answer:
The smallest aromatic species is cyclopropenyl cation.

Question 34. Write structural formulae of isomeric nitrotoluenes.
Answer:
Structural formulae ofisomeric nitrotoluenes are

Question 35. Write structural formulae of isomeric dibromophenols.
Answer:
Structural formulae of isomeric dibromo phenols are

Question 36. More than three dibromobenzenes are not possible—explain.
Answer:
Considering the resonance structures of benzene, it is easy to understand that positions 1,2- and 1,6- are indistinguishable. Similarly, positions 1,3- and 1,5- are indistinguishable. Thus, in the case of bromobenzene, only three isomers are possible which are as follows

Question 37. Write the IUPAC names of the given compounds

Answer:
- 1,2-dihydroxybenzene
- 1-phenylpropanoid-l-one
- 2-hydroxybenzoic acid
- Al-phenylethylamine
- l-bromo-3-chlorobenzene
- 3-phenylpropanoid acid
- 2,4,6-trinitrotoluene
- 4-hydroxy-3-methoxy benzaldehyde
Question 38. Write structures and IUPAC names:
- Mesitylene
- Styrene
- Pyrogallol
- Picric acid
- Salicylaldehyde
- Benzophenone
- TNT
- Phthalic acid
- Anthranilic acid.
Answer:

Question 39. Classify the following groups as o-/p-or m- directing group and activating or deactivating group:
- -NO2
- -Cl
- -C2H5
- -CP3
- OH
- — NHCOCH3
- —NH3
- — O
- —COCH3
Answer:
- — NO2 (deactivating and m -directing),
- —Cl (deactivating and o/p -directing),
- —C2H5 (activating and o-lp- directing),
- — CF3 (deactivating and m -directing),
- —OH (activating and o-lp- directing),
- — NHCOCH3 (activating and o-lp- directing),
- — NH3 (deactivating and m directing),
- — Oe (activating and o-lp-directing
- — COCH3 (deactivating and m -directing).
Question 40. Explain each of the following observations:
- Although —Cl is a deactivating group, it is o-lp-directly.
- The —CH3 group is an o-/p- directing group, even though the carbon atom contains no unshared pair of electrons.
- The —OCH3 group is an activating and o-/p directing group.
- The — CCl3 group is a m -m-directing group, even though the carbon atom is not bonded to a more electronegative atom by a double or triple bond.
Answer:
3. —OCH3 group is an o-/p-directing group because an unshared pair of electrons on O-atom participate in resonance (+R -effect) and increase the electron density of the ring at ortho- and para-positions.
So, electrophiles (E) preferably enter the ortho- and para-positions. Due to an increase in electron density, the ring becomes more activated than the unsubstituted benzene towards an electrophilic substitution reaction. Thus, — OCH3 is an activating group.
4. —CCl3 is an electron-withdrawing group because of its -I effect which is attributed to the presence of three highly electronegative Cl -atoms. Consequently, it decreases the electron density of the benzene ring, especially at the ortho- and para-positions.
So, —CCl3 is a deactivating group which makes the ring less reactive towards electrophilic substitution and substitution occurs preferably at meta-position.
NCERT Solutions Class 11 Chemistry Chapter 13 Hydrocarbons Long Q&A
Question 41. How will you prepare benzene from the given compounds?
- C6H5COOH
- C6H5CMe3
- C6H5CH2Cl
- C3H5Br
Answer:

Question 42. Write two processes to convert C6H6 into C6H5D.
Answer:

Question 43. Write the  formed.
formed.
Answer:

Question 44. Why groups like — CHO, — NO2, — B(OR) — PBr3 and — SR2 act as meta-directing groups?
Answer:
Since the carbon atom of the:
- — The CHO group is bonded to the oxygen atom by a double bond, the nitrogen atom of the
- — The NO2 group is linked with the oxygen atom by a double bond, the boron atom of the
- —B (OR)3 group contains a vacant p -orbital and the phosphorus and sulphur atoms of the groups
—+ PBr3 and — +SR2-, have vacant d-orbitals, all of these groups reduce the electron densities of ortho- and para- positions by their -R effect. Consequently, the electron density at the metaposition becomes relatively higher and the electrophile preferably enters the meta-position. Thus, these groups behave as meta-directing groups
Question 45. Arrange the compounds in increasing order of their rate of nitration and give reason: Benzene, Toluene, Nitrobenzene, Hexadeuterobenzene (C6D6).
Answer:
The increasing order of rate of nitration of given compounds is nitrobenzene < benzene = hexadeuterobenzene < toluene. The electron-attracting nitro (— NO2) group decreases the electron density of the nitrobenzene ring and as a result, its nitration occurs at a rate slower than that of benzene.
On the other hand, the electron-repelling methyl (— CH3) group increases the electron density of the toluene ring and as a result, its nitration proceeds at a rate faster than that of benzene. Benzene and hexadeuterobenzene (C6D6) undergo nitration at the same rate because, in an aromatic electrophilic substitution reaction, cleavage of the C — H or C — D bond does not occur at the rate-determining step.
Question 46. A mixture of benzene and bromine solution remains unchanged for an indefinite period, but if an iron nail is added to the solution, bromination of benzene occurs rapidly—explain.
Answer:
Benzene is an aromatic compound having no ethylenic unsaturation. So benzene does not participate in additional reactions with bromine. Again, the substitution reaction of benzene does not take place with the poor electrophile bromine alone.
So a solution of bromine in benzene remains stable (f.e., unchanged) for an indefinite period. However, when an iron nail is added to the solution, bromination of benzene occurs to yield bromobenzene because iron then acts as a halogen carrier. The red solution of bromine becomes colourless
2Fe + 3Br2→2FeBr3; Br2 + FeBr3 → Br+ FeBr–
C6H6 + Br+ FeBr–4 →C6H5Br + HBr + FeBr3
Question 47. Write the monosubstituted compounds formed in each of the following reactions and state whether each reaction is faster or slower than that of benzene.
- Nitration of C6H5NHCOCH3,
- Bromination of C6H5CBr3
- Chlorination of C6H5CMe3
- Nitration of C6H5—C6H5
- Nitration of C6H5—COOCH3
- Sulphonation of C6H5CHMe2
- Nitration of C6H5CN,
- Bromination of C6H5I,
- Nitration of C6H5-C6H4C6H5
Answer:
1. p-O2NC6H4NHCOCH3 (for this compound nitration occurs faster than benzene).
2. m-BrC6H4CBr3 (for this compound bromination occurs slower than benzene)
3. p-ClC6H4CMe3 (for this compound chlorination occurs faster than benzene)
4. p-O2NC6H4C6Hg (for this compound nitration occurs faster than benzene)
5. m-O2NC6H4COOMe (for this compound nitration occurs slower than benzene)
6. p-HSO3C6H4CHMe2 (for this compound sulphonation occurs faster than benzene)
7. m-O2NC6H4CN (for this compound nitration occurs slower than benzene)
8. p- BrC6H4I (reaction occurs slower than benzene)
9.

(reaction occurs faster than benzene in the middle ring because it is attached to two activating — CgH5 groups on both sides.)
Question 48. Write three methods by which alkyl side chains can be introduced into the benzene ring
Answer:
The: methods by which alkyl side chains, can be introduced into the benzene ring are—
1. By using (a) CH3CH3X, AlCl3, CH2=CH2 HF and CH3CH2OH, BF3 or concentrated H2SO4 in Friedel-Crafts alkylation reaction.
2. By acylation of benzene using CH3COCI or (CH3CO)2O, AlCl3 followed by . Clemmensen reduction ![]() the formed ketone.
the formed ketone.
4. By reacting CH3CH2Br with Ph2CuLi according to Corey-House synthesis.
Question 49. Classify the following groups based on their orientation and reactivity:

Answer:
1,4 and 5 are activating and ortho-/para- directing groups. 3 and 6 are deactivating and meta-directing groups. 2 is a deactivating group (due to — NO2 ) and is ortho-/para directing >C=C<
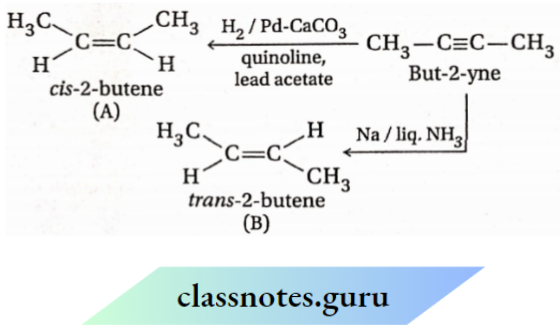
Question 50. 1-butyne and 2-butyne are allowed to react separately with the reagents given below:
- Na, liquid NH3 ;
- H2 (1 mole), Pd-BaSO4, quinoline,
- H2SO4,H2O, H2SO4 ;
- H2/Pt.
Which reagent(s) will produce the same product in both cases? Write the structures, of products formed in these cases
Answer:
Both 1-butyne & 2-butyne react separately with reagents 1 and 2 to produce 1-butene and 2-butene respectively
However, reagents(3 & 4 react with 1-butyne & 2-butyne separately to yield same products (2-butanone & butane)

Question 51. Ethyne reacts with dil. H2S04 in the presence of Hg2+ salts to give acetaldehyde, but with HC1, under similar conditions, it gives vinyl chloride. Account for such observation.
Answer:
In the first step, ethyne reacts with Hg2+ to form a cyclic complex (I). This is then attacked by more nucleophilic H2O, 2— rather than, weakly nucleophilic SO4, to form unstable vinyl alcohol which then tautomerism to give acetaldehyde

If HCl is used instead of H2SO4 then the complex (I) is attacked by more nucleophilic Cl-, rather than weakly nucleophilic H2O, to give vinyl chloride

An alkane has a molecular mass of 72. Give the structure of all the possible isomers along with their IUPAC OH names
Let the alkane be CnH2n+2. It’s molecular
=12n + (2n + 2) = 14n + 2
.*. 14n + 2 = 72, thus n – 5 and hence the alkane is C5H12
The isomers of the alkane C5H12 are—

Long Questions and Answers for Class 11 Chemistry Hydrocarbons
Question 52. Find the number of structural and configurational isomers of a bromo compound C5H9Br formed by the addition of HBR to 2-pentyne.
Answer:
Addition of one molar proportion of HBr to CH3—CH2—C=C—CH3 produces two structural isomers 1 and 2

Each of these structural isomers can exist as a pair of geometrical isomers (cis and trans) and hence there are four possible configurational isomers

Question 53. Identify the products P and Q in the following reaction:

Answer: In the absence of light, the reaction occurs via a polar mechanism

Question 54. Identify the product ‘T’ in the following reaction and the major product. Account for its formation.

Answer:
The product‘T is iodobenzene.
Explanation: Since I am less electronegative than Cl, so I+ is the effective electrophile that takes part in the reaction

Question 55. Identify the major product obtained on; monobromination (Br2/FeBr3) of meta methyl anisole and account for its formation
Answer:

Both —CH3 and —OCH3 are o-/p-directing groups. Therefore, the possible positions of attack which are facilitated by these groups are indicated by arrows as shown below

Attack by the electrophile (Br+) is disfavoured at C2 because this position is most crowded. Again -I effect of — the OCH3 group does not favour attack at C6. So most favourable attack occurs at C4, thereby producing 4-bromo-3-methylanhole as
Question 56. The enthalpy of hydrogenation of cyclohexene is -119.5 kj. mol-1 ,. If the resonance energy of benzene is 150.4 kj. mol-1 , estimate its enthalpy of Br2/FeBr3 hydrogenation.
Answer:
Enthalpy of hydrogenation of cyclohexene
=-119.5 kj- mol-1
So enthalpy of hydrogenation of hypothetical cyclohexatriene
= 3 × -119.5 kj- mol-1,
In other words, the calculated (or theoretical) enthalpy of hydrogenation of benzene =-3 × -119.5 kj- mol-1,
Let the actual (i.e., experimental) enthalpy of hydrogenation of benzene = × kj- mol-1
Now, R. E. of benzene = calculated enthalpy of hydrogenation of benzene- actual enthalpy of hydrogenation of benzene
o,-150.4 = -3 X 119.5 —x
x = -3 ×119.5 + 150.4 = -208.1 kj. mol-1
Question 57. How low will you prove:
- Acidic character of acetylene.
- Presence of terminal =CH2 group in 1-pentene.
- Presence of acetylenic hydrogen in 1-butyne.
- 2-butene is a symmetrical alkene. 1-butyne
Answer:
When acetylene is added to water and shaken, the resulting solution does turn blue litmus red. The following reactions in which the H-atoms of acetylene are replaced by metal atoms prove the acidic character of acetylene.
- HBr (a) Acetylene (HC=CH) reacts with sodium in two steps to form monosodium acetylide (HC = CNa) and disodium acetylide (NaC = CNa) respectively and in each case, H is evolved.

2. When acetylene gas is passed through ammoniacal Cu2Cl2 or AgNO3 solution, metallic acetylide is precipitated in each case
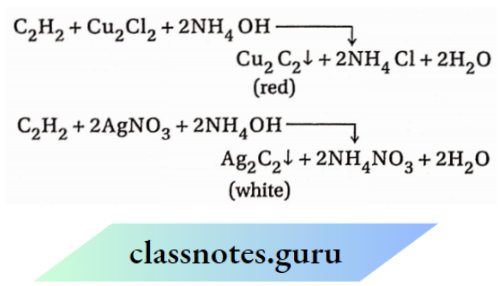
3. Ozonolysis of 1-pentene leads to the formation of formaldehyde (HCHO) as one of the products. This proves that there is a terminal =CH2 group present in 1-pentene

When 1-butyne is treated with ammoniacal cuprous chloride, a red precipitate of cuprous 1-butynide is obtained. Again, when an aqueous solution of silver nitrate is added to the alcoholic solution of 1-butyne, a white precipitate of silver 1-but nude is obtained.

Ozonolysis of any symmetrical alkene results in the formation of only one carbonyl compound (2 moles). Since 2-butene, on ozonolysis, produces two moles of acetaldehyde (CH3 CHO), it must be a symmetrical alkene. Its structure is CH3 CH=CH CH3.
Question 58. Identify the major product obtained in each of the following reactions and explain its formation

Answer:
1.

Although the alkene is an unsymmetrical one, Markownikoff’s rule is not directly applicable here because there are the same number of H-atoms attached to double-bonded carbons. Out of the 2 carbocations ( C6H5CHCH2CH3 & C6H5CH2CHCH3 ) obtained in the first step of the reaction, the first one (a benzylic carbocation) is more stable because it is stabilised by resonance involving the benzene ring. So, this carbocation is formed more easily and readily and in the second step, it combines with the Br ion to give 1-bromo-l-phenylpropane as the major product.
2.

This reaction occurs according to Markownikoff’s rule. Chlorine is more electronegative than iodine. So, in the Iδ+– Clδ- molecule, the I-atom with a partial positive charge combines first with the alkene as an electrophile. Between the two carbocations, (CH3)2+CCH2I and (CH3)2+CICH2 formed in the first one being a 3° carbocation is relatively more stable. So, it is formed more easily and readily and in the second step, it combines with Cl to give 2-chloro-lido-2-methylpropane as the major product.
3.

Double bond is more reactive than triple bond towards electrophilic addition reactions. For this reason, bromine (1 mole) is added mainly to the double bond of the compound to produce 4, 5 -dibromo pent-1-one as the major product.
Question 59. Write the names and structures of the two alkenes (molecular formula: C4H6) which give the compound when added to HBr in the absence of organic peroxide but different compounds when added to HBr in the presence of peroxide
Answer:
When but-1-ene (CH3CH2CH=CH2) and but-2-ene (CH3CH=CHCH3) react with HBr in the absence of an organic peroxide, the same product is obtained.

However, in the presence of an organic peroxide, the addition of HBr to but-1-ene occurs contrary to Markownikoff’s rule and hence differences are obtained from these two cases
1.

2.

Question 60. Mention two reactions in which ethylene and benzene behave differently and two reactions in which they behave similarly.
Answer:
Benzene, unlike ethylene, fails to discharge the red colour of bromine in CCl4 or the reddish-violet (purple) colour of potassium permanganate solution because, unlike ethylene the π -electron system of benzene possesses extraordinary.
O2 stabilisation.
So, in these two reactions ethylene and benzene behave differently.

In the following two reactions, ethylene and benzene behave similarly.
1. Both ethylene and benzene bums with sooty flame to produce CO2 and H2O
⇒ \(\mathrm{C}_2 \mathrm{H}_4+3 \mathrm{O}_2 \longrightarrow 2 \mathrm{CO}_2+2 \mathrm{H}_2 \mathrm{O}\)
⇒ \(2 \mathrm{C}_6 \mathrm{H}_6+15 \mathrm{O}_2 \longrightarrow 12 \mathrm{CO}_2+6 \mathrm{H}_2 \mathrm{O}\)
2. Both ethylene and benzene react with ozone to form ozonide (an additional
⇒ \(\mathrm{C}_2 \mathrm{H}_4+\mathrm{O}_3 \longrightarrow \mathrm{C}_2 \mathrm{H}_4 \mathrm{O}_3\) (Ethylene (ozonides)
⇒ \(\mathrm{C}_6 \mathrm{H}_6+3 \mathrm{O}_3 \longrightarrow \mathrm{C}_6 \mathrm{H}_6 \mathrm{O}_9\) (Benzene tri ozonide)
Question 61. Distinguish between:

Answer:
1. Toluene (C6H5CH3), on oxidation by alkaline KMnO4 solution followed by acidification gives a white crystalline precipitate of benzoic acid (C6H5COOH). On the other hand, tert-butylbenzene does not undergo such an oxidation reaction by an alkaline KMnO4 solution.

2. O -xylene and m -xylene, on oxidation by alkaline KMnO4 solution, produce phthalic acid and isophthalic acid respectively. Phthalic acid, when heated, forms phthalic anhydride which responds to phthalein test On the other hand, isophthalic acid on heating does not produce any anhydride.

Hydrocarbons Chapter 13 Long Question and Answers Class 11
Question 62. Write the name & structure of the following compounds:
- An unsaturated aliphatic hydrocarbon which forms monosodium salt.
- An organic compound which causes depletion of the ozone layer.
- An alkane which is used as a fuel for household cooking.
- An alkyl bromide which reacts with alcoholic KOH to form only 1-butene.
- An alkene which reacts with HBr in the presence or absence of peroxide to give the same product.
- A compound containing iodine which, when heated with silver powder, produces acetylene.
- An alkyl bromide (C4H9Br) does not participate in the Wurtz reaction.
- An alkene which on ozonolysis forms glyoxal and formaldehyde.
Answer:
- Propyne (CH3C= CH).
- Dichlorodifluoromethane (CF2Cl2).
- Butane(CH3CH2CH2CH3).
- 1-bromobutane (CH3CH2CH2CH2Br).
- 2-butene (CH3CH=CHCH3).
- Iodoform (CHI3).
- Tert-butylbromide (Me3CBr).
- 1,3-butadiene (CH2=CH—CH=CH2)
Question 63. Identify A … G in the following reaction sequence

Answer:

Question 64. Write structures and names of the compounds A to Q in the following reaction sequences:

Answer:


Question 65. How will you distinguish between each of the given pairs of compounds by a single chemical test?
- Ethylene and acetylene
- Ethane and acetylene
- 1-butyne and 2-butyne
- Ethane and ethylene.
- Propene and propyne
- 1-butene and 2-butene
- 2-pentene and benzene
- Benzene and cyclohexene.
Answer:
The distinction between two compounds should be written in a tabular form. A reagent which either causes a colour change, or evolution of a gas or the appearance of a precipitate should be selected for this purpose.



Question 66. How will you carry out the following transformations:
- Acetylene → Acetone
- Acetylene →Dldeuteroacetylene(C2D2)
- Acetylene → Acetylenedicarboxyllc acid
- 1-butyne → 2-butyne
- Propene → 1-propanol Propyne → Propanal
Answer:


Question 67. The boiling points of three isomeric pentanes are 36.2°C, 28°C and 9.5°C respectively. Identify the compounds and give a reason.
Answer:
The strength of van der Waals forces depends on the area of products, A and B obtained in the following reactions: contact between molecules. The area of contact between straight-chain n -pentane (CH3CHCH2CH2CH3) molecules is maximum. So, the extent of van der Waals’ attraction among its molecules is maximum. For this reason, its boiling point is highest (36.2°C). On the other hand, the area of contact between spherical neopentane [(CH3)4C] molecules is minimal.
So, _ the extent of van der Waals’ attraction among its molecules is the minimum. For this reason, its boiling point is the lowest (9.5°C). Again, the area of contact between isopentane [(CH3)2CHCH2CH3] molecules is intermediate between n-pentane and neopentane and so, its boiling point (28°C) is intermediate between the other two isomers.
Class 11 Hydrocarbons Long Questions and Answers NCERT Solutions
Question 68. Write the structure and the name of the monobromoderivative which is obtained as the major product when n-butane reacts with bromine in the presence of light. Why is it produced in larger amounts?
Answer:
N-butane reacts with bromine in the presence of light to give 2-bromobutane as the major product.

The reaction occurs through the free radical mechanism. As 2° free radical (CH3CH2CHCH3) is relatively more stable than 1° free radical (CH3CH2CH2CH2), displacement of 2° H-atom occurs rapidly to give 2-bromobutane as the major product
Question 68. Possible methods for the preparation of 4-methyl-2- pentyne arc are given. Which method is desirable & why?

Answer:
In both the methods given, the reaction in the second step (reddish-brown) proceeds through the SN2 pathway and it is known that an SN2 reaction is very susceptible to steric effect So, the product will be obtained in good yield if, in the second step, methyl or primary alkyl bromide is used. In the second step of the second method, methyl bromide (CH3Br) has been used. Hence, the second method is desirable.
Question 70. Three separate cylinders contain methane, and ethylene acetylene respectively. How will you identify them?
Ana.
The three gases are first separately passed through the ammoniacal solution of cuprous chloride. The gas, which gives a red precipitate, is acetylene. The gases in the remaining two cylinders are separately passed through a solution of bromine in CCl4. The gas, which decolourises the reddish-brown solution of bromine, is ethylene. Hence, the remaining gas in the other cylinder is methane.

Question 71. Give an example alkene which on oxidation by acidic solution of KMn04 or on ozonolysis gives the same compound. Give reason.
Answer:
A terminal =CR2 group of an alkene gets converted into a ketone when the alkene is heated with an acidic solution of KMnO4 or subjected to ozonolysis. Hence, an example of such an alkene is 2,3-dimethyl but-2-ene

Question 72. Write the formulas and names of the alkenes which on hydrogenation form 2-methylpentane.
Answer:
The carbon skeleton of the probable alkenes is

As there are four different positions of the double bond in the given carbon skeleton, four alkenes are possible which form 2-methylpentane on hydrogenation. The probable alkenes are:

Question 73. Write two possible methods of preparing 2-methylpropane by Corey-House synthesis. Out of these two methods, which one is better and why?
Answer:
Two possible methods of preparing 2-methylpropane by Corey-House synthesis are as follows-

In Corey-House synthesis, the third step is an SN2 reaction (sensitive to steric effect). So, this step is highly favourable for methyl or primary halides, and less’ favourable for secondary alkyl halides arid1 does not occur in the case of tertiary alkyl halides. In methods (1) and (2), a secondary halide and methyl halide have been used respectively In ease of the third step. So, method
(1) Is better than the method
(2) for preparing 2-methyl propane by Corey-House synthesis.
Question 74. A or B  A and B are the two geometrical isomers. Identify them.
A and B are the two geometrical isomers. Identify them.
Answer:
The alkene which gives only acetaldehyde on ozonolysis is 2-butene (CH3– CH —CH CH3).
CH3CH=O+O=CHCI ⇒ CH3CH=CHCH3
So, A and B are the two geometrical isomers of 2-butene:

Question 75. Dlazomethane (CH2N2), on decomposition forms singlet methylene (: CH2) which gets attached to different non-equivalent C—H bonds of alkanes to form various alkanes. Name the alkanes formed when pentane (CH3CH2CH2CH2CH3) reacts with singlet methylene. Assuming methylene to be highly reactive and less selective, calculate the probable amounts of the formed alkanes.
Answer:
Three alkanes are formed when pentane reacts with singlet methylene because there are three non-equivalent C—H bonds in pentane molecules. So, the alkanes formed are:

As methylene is highly reactive and less selective, its insertion occurs randomly. So, the amounts of the formed compounds have calculated the basis of the probability factor and number of equivalent C— H bonds. For example, Percentage of hexane,
(CH3CH2CH2CH2CH2CH3) = \(\frac{6}{12}\) × 100 = 50
Percentage of 2-methyl pentane
CH3—CH – CH3—CH2CH2CH3 =\(\frac{6}{12}\) × 100 = 50
percentage of 3-methyl pentane = \(\frac{2}{12}\) × 100 = 16.7
Question 76. How will you prepare (CH3)2CD14 CH3 from propane (CH3CH2CH3)?
Answer:

NCERT Class 11 Chemistry Hydrocarbons Long Question and Answer PDF
Question 77. How will you prepare \({ }^{14} \mathrm{CH}_3{ }^{14} \mathrm{CH}_2{ }^{14} \mathrm{CH}_3\) taking \({ }^{14} \mathrm{CH}_3 \mathrm{I}\) as the only source of carbon?
Answer:

Question 78. In the reaction of 2-pentene with HI, the two isomeric iodopentanes are produced in almost equal amounts —why?
Answer:
The two doubly bonded carbon atoms in 2-pentene are bonded to the same number (one) of H-atoms. So, the two isomeric iodopentanes are produced in nearly equal amounts.

The two 2° carbocations (CH3CH2CH2CHCH3 and CH3CH2CHCH2CH3 ) obtained on the addition of proton at C-2 or C-3 are almost equally stable. So, the reaction proceeds through the two routes nearly at the same rate and consequently, the two isomeric iodopentanes are formed in nearly equal amounts.
Question 79. From the following two reactions, arrange HC = CH, NH3 and H2O in the increasing order of their acidic character.
- HC = CH + NaNH2→ HC = CNa + NH3
- HC = CNa + H2O → HC = CH + NaOH
Answer:
In reaction no. , HC ≡ CH exhibits its acidic character and produces NH3 from NaNH2. So HC = CH is more acidic than NH3.
On the other hand, in reaction (2), water exhibits its acidic character and produces HC = CH from HC = CNa. So, H2O is more acidic than HC = CH. Thus, the increasing order of acidic character: NH3 < HC=CH < H2O
Question 80. Unlike acetylene, ethylene dissolves in concentrated sulphuric acid—why?
Answer:
In the first step of the reaction with concentrated H2SO4, ethylene forms an ethyl cation (CH3C+H2) and acetylene forms a vinyl cation (CH2 =+CH) by accepting a H® ion.
Since vinyl cation is less stable than ethyl cation, in the case of acetylene, the first step (rate-determining step) of the reaction does not occur easily. Thus, unlike ethylene, acetylene fails to dissolve in concentrated H2SO4
Question 81. Write the structure of the product expected to be formed when CH2=CH—CH3(C = 14C) is subjected to free radical chlorination.
Answer:

Question 82. Identify the products obtained when ethylene gas is passed through bromine water in the presence of sodium chloride.
Answer:

Question 83. Which alkenes are formed by dehydrating the following alcohols in the presence of acid? Give the reaction mechanism.

Answer:

Question 84. The conjugated dienes are more reactive than alkenes which in turn are more reactive than alkynes towards electrophilic addition reactions —explain.
Answer:
The reactivity of alkenes, alkynes or conjugated dienes towards electrophilic addition reaction depends on the stability of the intermediate carbocation obtained in the rate-determining step by addition of the electrophile (E+).

Out of the three carbocations (la, Ila and IUa), (IlIa) is the most stable because it is stabilised by resonance. Again, out of (a) and (Ila), (IIla) is less stable because the positive charge in it is placed on a more electronegative sp2 -hybfiflis6d carbon atom.
Thus, the stabilities of these carbocations follow the order IIIa> la > Ila. Therefore, the order of activity of these compounds is conjugated diene > alkene > alkyne.
Chapter 13 Hydrocarbons NCERT Solutions Long Questions Class 11
Question 85. Calculate the resonance energy of 1,3-butadiene from the following data

Answer:
The heat liberated due to hydrogenation of one double bond = 30 kcal – mol-1
The heat liberated due to hydrogenation of two double bonds = 30 × 2 = 60 kcal – mol-1.
Heat liberated due to hydrogenation of 1,3-butadiene (CH=CH—CH=CH2) = 57 kcal -mol-1.
Therefore, resonance energy of 1,3-butadiene = 60- 57 = 3 kcal – mol-1
Question 86. Dehydration of alcohols to alkene is carried out by treating with a cone. H2SO4 but not with cone. HCl or HNO3. Give reasons.
Answer:
Dehydration of alcohol proceeds via the formation of a carbocation intermediate. If HCl is used as the dehydrating agent then chloride ion (Cl–), being a good nucleophile, attacks at carbonium ion carbon (Cl+) thereby producing alkyl chloride as the substitution product together with the alkene as the elimination product.

If cone. H2SO4 Is used as the reagent the H2SO4 ion derived from H2SO4 does not act as a nucleophile. Instead, the carbocation loses a proton from the β -carbon atom to give alkene (R—CH=CH2) as the elimination product.

If cone. HNO3 is used as the reagent then it being a strong oxidising agent, brings about oxidation of the alcohol first to an aldehyde or a ketone and then to a carboxylic acid.

Question 87. How will you prepare ethylbenzene by using ethyne as the only organic substance and any other inorganic substance of your choice?
Answer:
Ethylbenzene (C6H5C2H5) may be prepared from ethyne (acetylene) through the following steps:

Question 88. Explain why the bromination of benzene requires FeBr3 as a catalyst, while the bromination of anisole (C6H5OCH3) does not require any catalyst.
Answer:
Since the benzene molecule is not so reactive,© for bromination it requires more reactive bromine cation (Br) or the complex Br— Br—FeBr3 as the electrophile. Due to the presence of electron-donating (+R) methoxy (— OCH3) group.
The anisole ring becomes much more reactive towards an electrophilic substitution reaction. When the non-polar bromine molecule comes in contact with the anisole ring, it 6+ 6 — becomes partially polarised (Br— Br)and its positive end (weak electrophile) undergoes easy attack by anisole. Therefore, due to the greater reactivity of anisole, its bromination requires no catalyst.
Question 89. Neither vinyl chloride (CH2=CH—Cl) nor chlorobenzene (C6H5 —Cl) can be used as an alkylating agent in the Friedel-Crafts reaction—why?
Answer:
In vinyl chloride or chlorobenzene, the unshared pair of electrons on the Cl atom is involved in resonance interaction with the σ -electron system and as a result, the C—Cl bond in both cases acquires some double bond character.
The Lewis acid AlCl3 is incapable of breaking such a strong C—Cl bond. Moreover, even if the C— Cl bond breaks, the carbocations produced would be unstable (due to a positive charge on an sp² – hybridised carbon atom).
Hence such a bond is very much reluctant to undergo cleavage. For this reason, vinyl chloride or chlorobenzene cannot be used as an alkylating agent in the Friedel-Crafts reaction.
Question 90. Two methods for the preparation of propylbenzene are given below-

Question 91. Which one of the two methods is more effective for the preparation of propylbenzene? Give reason.
Answer:
Method 2 is more effective for the preparation of propylbenzene. This is because, in method 1, the alkylating agent containing a chain of three carbon atoms isomerises to give isopropyl benzene as the principal product.

Moreover, the alkyl group activates the benzene ring towards further substitution. So, there is a possibility of polyalkylation of benzene. However, although method 2 involves two steps, the desired propylbenzene is obtained as the only product with a higher yield. In the first step of the reaction.
The CH3CH2CO — group is introduced into the ring. Since the acyl group has no possibility of isomerisation, no other isomeric group can enter the ring.
Furthermore, the acyl group being an electron-attracting one deactivates the ring and consequently, polyacylation cannot take place. Hence in the first step, only propiophenone (C6H5COCH2CH3) is produced and in the second step, it is reduced by Clemmensen method to give only propylbenzene.
Question 92. Which ring (A or B) is in each of the following? Compounds will undergo nitration readily and why?
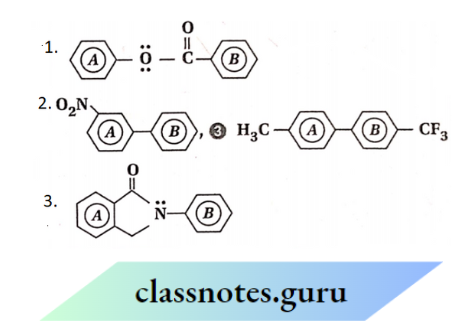
Answer:
- Ring A is attached to the electron-donating or activating group — :O: COPh, whereas ring B is attached to the electron-attracting or deactivating — COOPh group. So, ring A is more reactive than ring B towards electrophilic substitution reaction. Hence, ring A will undergo nitration readily.
- Since a deactivating — N02 group is attached to ring A, it is relatively less reactive than ring B towards electrophilic substitution. Consequently, ring B undergoes nitration readily.
- Ring A is attached to an electron-donating or activating — CH3 group while ring B is linked to an electron-attracting or deactivating — CF3 group. So, ring A is relatively more reactive than ring B towards electrophilic substitution. Hence, ring A undergoes nitration at a faster rate.
- Ring B undergoes nitration readily. The reason is similar to that given in the case of compound (1).
Question 93. Write the names and structures of the compounds formed during the Friedel-Crafts reaction of benzene with
- CH2Cl2
- CHCl3 and
- CCl4
Answer:

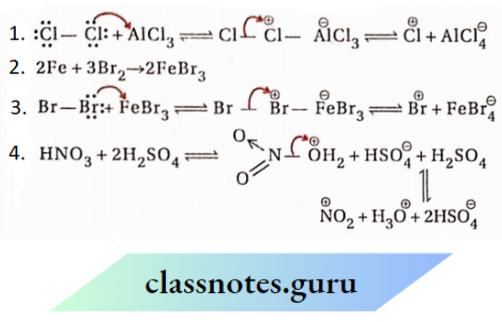
Question 94. Show the formation of the electrophile in each case:
- Cl3/AlCl3
- Br2/Fe
- Conc.HNO3 + conc.H2SO4
Answer:
Question 95. How will you prepare the following compounds from benzene?
- PhCH2CH2Ph,
- PhCH2CH2CH2Ph and
- PhCH2CH2CH2CH2Ph
Answer:

Question 96. An optically active compound A (C10H4) gets oxidised to benzoic acid (C6H5COOH) by alkaline KMnO4 However, compound B, which is an optically inactive isomer of A does not get oxidised by alkaline KMn04. Identify A and B.
Answer:
As A is oxidised to C6H5COOH, A is a substituted benzene which has only one side chain consisting of four carbon atoms. Again, as A is optically active, there must be an unsymmetric carbon atom present in the side chain.
So, the side chain is —CH(CH3)CH2CH3 and A is sec-butylbenzene, C6H5CH(CH3)CH2CH3. B, an isomer of A does not get oxidised by alkaline KMnO4.
Thus, there is no benzylic hydrogen in the compound. So, the side chain is — C(CH3)3. The compound B is tert-butylbenzene, C6H5C(CH3)3
Question 97. Considering the stability of <r -complex, explain why — OCH3 is o-lp- orienting while —NO2 is mega-orienting.
Answer:
Electrophilic substitution reaction in anisole proceeds via the following reaction mechanism:

There is an extraordinarily stable (every atom has its octet fulfilled) resonance structure in both ortho- and para-a -complex, but there is no such resonance structure in the meta-σ- complex. So, ortho- and para-cr -complex is more stable than meta-cr complex. Consequently, electrophilic substitution proceeds easily and rapidly via ortho- and para-a -complex resulting in ortho and para-substituted compounds as major products. Thus, — OCH3 is ortho-/para-orienting group.
Electrophilic substitution reaction in nitrobenzene proceeds via the following reaction mechanism:

Both ortho- and para-complexes are extraordinarily unstable resonance structures (due to the presence of a positive charge on two adjacent atoms). However, in meta-σ -complex there is no such resonance structure and so it is more stable than ortho- and para- σ -complex.
Thus, the reaction proceeds rapidly via the meta- σ -complex and the meta- substituted compound is obtained as the major product Thus, — NO is /nefa-orienting group.
Question 98. Identify (A)-(F) in the following reaction

Answer:

Question 99.
1. What will be the major product when propyne is treated with aqueous H2SO4? Explain the equation.
2. An organic compound (A), C7H8O is insoluble in aqueous NaHCO3 but soluble in NaOH. (A), on treatment with bromine water rapidly forms compound (B), C7H5OBr2. Give structures of (A) & (B). What will be (A) if it does not dissolve in NaOH solution but shows the reaction given above?
Answer:
Propyne does not react with aqueous H2SO4 in the absence of Hg2+ ion. In the presence of an Hg2+ ion, propyne reacts with aqueous H2SO4 to give the unstable compound prop-2-enol (according to Markownikoff’s rule) which tautomerism to give acetone.

The problem is solved by assuming that the compound ‘B’ has the molecular formula C7H6OBr2

Long Answer Questions on Hydrocarbons Class 11 NCERT
Question 100. Write the structural formula of the compounds A to F:

Answer:

Question 101. Both Br2(g) and NO2(g) are reddish-brown gaseous substances. How will you chemically distinguish between them?
Answer:

Question 102. Draw the structural formula of the compound from A to F.
Answer:

Question 103. Convert:
- 2-propanol → 1-propanol
- 2-butene→Ethane
Answer:

Question 104. Write the IUPAC names of the following compounds:

Answer:

Question 105. For the Riven compounds write structural formulas and IUFPAC names for all possible isomers having the number of double or triple bonds as indicated:
- C4H8 (one double bond)
- C5H8 (one triple bond)
Answer:

Question 106. Write IUPAC names of the products obtained by the ozonolysis of the following compounds:
- Pent-2-ene
- 3,4-dimethyIhept-3-ene
- 2-ethyl but-1-ene
- I-phenyl but-1-ene
Answer:

Question 107. Explain why the following systems are not aromatic.
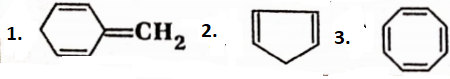
Answer:
1.

There are no p-orbitals on one of the H H CH3H CH3 carbon atoms forming the ring structure of this system and It is not a cyclic conjugated polyene containing (4n + 2)n -electrons. So, the system is not aromatic.
2.

There are no p-orbitals on one of the carbon atoms forming the ring structure of this system and it is not a cyclic conjugated polyene containing (4n + 2)n -electrons. So, the system is not aromatic.
3.
 Cyclooctatetraene has a non-planar structure and there are 8π -electrons in it. So, cyclooctatetraene is a non-aromatic compound.
Cyclooctatetraene has a non-planar structure and there are 8π -electrons in it. So, cyclooctatetraene is a non-aromatic compound.
Question 108. How will you convert benzene into
- p-nitrobromobenzene
- m-nltrochlorobenzene
- p-nitrotoluene
- Acetophenone
Answer:

Hydrocarbons Class 11 Long Question and Answers NCERT
Question 109. In H3C—CH2—C(CH3)2—CH2CH(CH3)2, identify 1°, 2°, and 3° carbon atoms and give the number of H atoms bonded to each one of these.
Answer:

Number of H-atoms attached to 1° carbon atom = 15
Number of H-atoms attached to 2° carbon atom = 4
Number of H-atoms attached to 3° carbon atom = 1
Question 110. Write down the products of ozonolysis of 1,2-dimethylbenzene (o-xylene). How does the result support the Kekule structure for benzene?
Answer:

As the products A, B and C cannot be obtained from any one of the two Kekule structures, this confirms that o-xylene is a resonance hybrid of the two Kekule structures 1 and 2.

Question 111. Arrange benzene, n-hexane and ethyne in decreasing order of acidic behaviour. Also, give a reason for this behaviour.
Answer:
The hybridisation state of carbon in the compounds benzene, n-hexane and ethyne is as follows—

The nucleus. Thus, the correct order of decreasing acidic behaviour is ethyne > benzene > n-hexane.
Question 112. How would you convert the given compounds into benzene?
- Ethyne
- Ethene
- Hexane
Answer:

Question 113. Write structures of all the alkenes which on hydrogenation give 2-methylbutane.
Answer:
The structural formula of 2-methyl butane is—

The structures of different alkenes by putting double bonds at different positions along with satisfying the tetravalency of each carbon atom which gives 2-methyl butane on hydrogenation are as follows-

Question 114. Arrange the following set of compounds in order of their decreasing relative reactivity with an electrophile, E+.
- Chlorobenzene, 2,4-dinitrochlorobenzene, p-nitrochlorobenzene
- Toluene, p-H3C —C6H4—NO2, P-O2N—C6H4—NO2
Answer:
The electron density of the benzene nucleus increases in the presence of an electron-donating group (activating group). Consequently, electrophiles can easily attack the benzene nucleus. On the other hand, the electron density of the benzene nucleus decreases in the presence of the electron-withdrawing group (deactivating group). This makes electrophilic substitution difficult for the benzene nucleus.
Therefore, the order of the different compounds according to their decreasing relative reactivity with an electrophile E+ is—
Chlorobenzene > p-nitrochlorobenzene > 2,4-dinitrochlorobenzene
Toluene > p-CH3C6H5NO2 > p-O2NC6H4NO2
