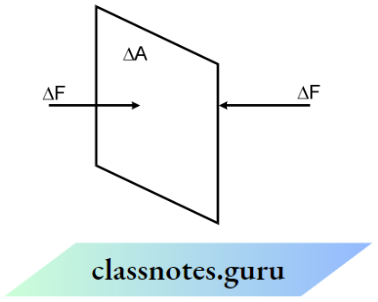
Kinetic Theory Of Gases And Thermodynamics
Kinetic Theory Of Gases:
The kinetic theory of gases is based on the following basic assumptions.
- A gas consists of a very large number of molecules. These molecules are identical, perfectly elastic, and hard spheres. They are so small that the volume of molecules is negligible compared with the volume of the gas.
- Molecules do not have any preferred direction of motion, motion is completely random.
- These molecules travel in straight lines and free motion most of the time. The time of the collision between any two molecules is very small.
- The collision between molecules and the wall of the container is perfectly elastic. It means kinetic energy is conserved in each collision.
- The path traveled by a molecule between two collisions is called the free path and the mean of this distance traveled by a molecule is called the mean free path.
- The motion of molecules is governed by Newton’s law of motion
- The effect of gravity on the motion of molecules is negligible.
Kinetic Theory of Gases and Thermodynamics NEET Class 11 Notes
Expression For The Pressure Of A Gas
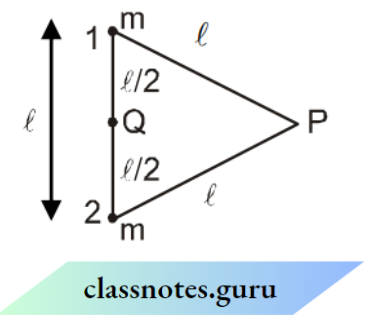
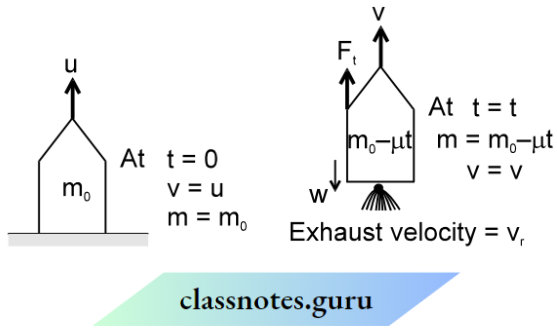
Let us suppose that a gas is enclosed in a cubical box having length l. Let there be ‘ N ‘ identical molecules, each having mass ‘ m’ since the molecules are of the same mass and perfectly elastic, so their mutual collisions result in the interchange of velocities only.
Only collisions with the walls of the container contribute to the pressure of the gas molecules. Let us focus on a molecule having velocity v1 and components of velocity vx1, vy1, and vz1 along the x, y, and z-axis as shown in the figure.
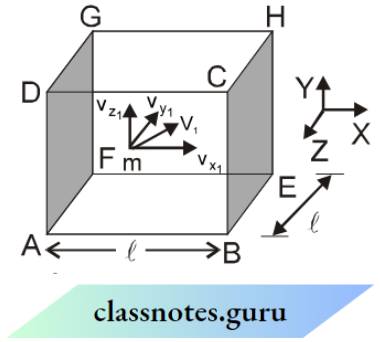
The change in momentum of the molecule after one collision with wall BCHE
⇒ \(m v_{x_1}-\left(-m v_{x_1}\right)=2 m v_{x_1}\)
The time taken between the successive impacts on the face BCHE = \(\frac{\text { distance }}{\text { velocity }}\)
⇒ \(\frac{2 \ell}{v_{\mathrm{x}_1}}\)
Time rate of change of momentum due to collision = \(\frac{\text { change in momentum }}{\text { time taken }}\)
⇒ \(\frac{2 m v_{\mathrm{x}_1}}{2 \ell / \mathrm{v}_{\mathrm{x}_1}}=\frac{\mathrm{mv}_{\mathrm{x}_1}^2}{\ell}\)
Hence the net force on the wall BCHE due to the impact of n molecules of the gas is :
⇒ \(\mathrm{F}_{\mathrm{x}}=\frac{\mathrm{mv}_{\mathrm{x}_1}^2}{\ell}+\frac{\mathrm{mv}_{\mathrm{x}_2}^2}{\ell}+\frac{\mathrm{mv}_{\mathrm{x} 3}^2}{\ell}+\ldots \ldots \ldots .+\frac{\mathrm{mv}_{\mathrm{x}_{\mathrm{n}}}^2}{\ell}\)
⇒ \(\frac{m}{\ell}\left(v_{x_1}^2+v_{x_2}^2+v_{x_3}^2+\ldots \ldots \ldots \ldots+v_{x_n}^2\right)\)
⇒ \(\frac{\mathrm{mN}}{\ell}<\mathrm{v}_{\mathrm{x}}^2>\)
where < V2x > = mean square velocity in x-direction. Since molecules do not favor any particular direction therefore
⇒\(\left\langle\mathrm{v}_{\mathrm{x}}^2\right\rangle=\left\langle\mathrm{v}_{\mathrm{y}}^2\right\rangle=\left\langle\mathrm{v}_{\mathrm{z}}^2\right\rangle\)
But
⇒\(\left\langle\mathrm{v}^2\right\rangle=\left\langle\mathrm{v}_{\mathrm{x}}^2\right\rangle+\left\langle\mathrm{v}_{\mathrm{y}}^2\right\rangle+\left\langle\mathrm{v}_{\mathrm{z}}^2\right\rangle\)
⇒ \(\left\langle\mathrm{v}_{\mathrm{x}}^2\right\rangle\)
⇒ \(\frac{\left\langle\mathrm{v}^2\right\rangle}{3}\) Pressure is equal to force divided by area.
P = \(\frac{\mathrm{F}_{\mathrm{x}}}{\ell^2}=\frac{\mathrm{M}}{3 \ell^3}\left\langle\mathrm{v}^2\right\rangle=\frac{\mathrm{M}}{3 \mathrm{~V}}\left\langle\mathrm{v}^2\right\rangle\).
Pressure is independent of x, y, and z directions.
Where l3 = volume of the container = V
M = total mass of the gas, <c2 > = mean square velocity of molecules
⇒ \(P=\frac{1}{3} \rho\left\langle v^2\right\rangle\)
As PV = nRT , then total translational K.E. of gas = \(\frac{1}{2} M<v^2>=\frac{3}{2} P V=\frac{3}{2} n R T\)
Translational kinetic energy of 1 molecule = \(\frac{3}{2} \mathrm{kT}\)(it is independent of nature of gas) kT
⇒ \(\left\langle v^2\right\rangle=\frac{3 P}{\rho}\)
or \(v_{rm s}=\sqrt{\frac{3 P}{\rho}}=\sqrt{\frac{3 R T}{M_{\text {mole }}}}=\sqrt{\frac{3 k T}{m}}\)
Where vrms is the root mean square velocity of the gas.
Pressure exerted by the gas is P = \(\frac{1}{3} \rho\left\langle v^2\right\rangle=\frac{2}{3} \times \frac{1}{2} \rho<v^2>\)
or \(P=\frac{2}{3} E, E=\frac{3}{2} P\)
Thus, the total translational kinetic energy per unit volume (it is called energy density) of the gas is numerically equal to \(\frac{3}{2} k T\) times the pressure exerted by the gas.
Important Points
- \(\mathrm{v}_{\mathrm{rms}} \propto \sqrt{\mathrm{T}} \text { and } \mathrm{v}_{\mathrm{rms}} \propto \frac{1}{\sqrt{\mathrm{M}_{\text {mole }}}}\)
- At absolute zero, the motion of all molecules of the gas stops.
- At higher temperatures and low pressure or higher temperatures and low density, a real gas behaves as an ideal gas.
Maxwell’s Distribution Law
Distribution Curve – A plot of \(\frac{\mathrm{dN}(\mathrm{v})}{\mathrm{dv}}\)(number of molecules per unit speed interval) against c is known as Maxwell’s distribution curve. The total area under the curve is given by the integral.
⇒ \(\int_0^{\infty} \frac{d N(v)}{d v} d v=\int_0^{\infty} d N(v)=N\)
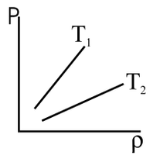
- Figure shows the distribution curves for two different temperatures. At any temperature, the number of molecules in a given speed interval dv is given by the area under the curve in that interval (shown shaded).
- This number increases, as the speed increases, upto a maximum and then decreases asymptotically toward zero.
- Thus, a maximum number of the molecules have speed lying within a small range centered about the speed corresponding to the peak (A) of the curve. This speed is called the ‘most probable speed’ vP or vmp. dN(v)

The distribution curve is asymmetrical about its peak (the most probable speed vP) because the lowest possible speed is zero, whereas there is no limit to the upper speed a molecule can attain. Therefore, the average speed v is slightly larger than the most probable speed vP. The root-mean-square speed, vrms, is still larger
⇒ \(\left(v_{\text {rms }}>\bar{V}>v_p\right)\)
Average (or Mean) Speed:
⇒ \(\bar{v}=\sqrt{\frac{8}{\pi} \frac{k T}{m}}=1.59 \sqrt{\mathrm{kT} / \mathrm{m}}\) (derivation is not in the course)
RMS Speed:
⇒ \(v_{\mathrm{rms}}=\sqrt{\left\langle v^2\right\rangle}=\sqrt{\frac{3 \mathrm{kT}}{\mathrm{m}}}=1.73 \sqrt{\frac{\mathrm{kT}}{\mathrm{m}}}\)
Most Probable Speed:
The most probable speed vP or vmp is the speed possessed by the maximum number of molecules and corresponds to the maximum (peak) of the distribution curve. Mathematically, it is obtained by the condition.
⇒ \(\frac{d N(v)}{d v}=0\)[by substitution of formula of dN(v) (which is not in the course)]
Hence the most probable speed is
⇒ \(v_p=\sqrt{\frac{2 k T}{m}}=1.41 \sqrt{\mathrm{kT} / \mathrm{m}}\)
From the above expression, we can see that
⇒\(\mathrm{v}_{\mathrm{rms}}>\overline{\mathrm{v}}>\mathrm{v}_{\mathrm{p}}\)
Degree Of Freedom
A total number of independent coordinates that must be known completely specify the position and configuration of a dynamical system completely is known as “degree of freedom f”. The maximum possible translational degrees of freedom are three i.e.
⇒ \(\left(\frac{1}{2} m V_x^2+\frac{1}{2} m V_y^2+\frac{1}{2} m V_z^2\right)\)
The maximum possible rotational degrees of freedom are three i.e.
⇒ \(\left(\frac{1}{2} I_x \omega_x^2+\frac{1}{2} I_y \omega_y^2+\frac{1}{2} I_z \omega_z^2\right)\)
Vibrational degrees of freedom are two i.e. (Kinetic energy. of vibration and Potential energy of vibration)
Mono Atomic: (all inert gases, He, Ar, etc.) f = 3 (translational)
Diatomic: (gases like H2, N2, O2 etc.) f = 5 (3 translational + 2 rotational)
If temp < 70 K for diatomic molecules, then f = 3
If the temp is between 250 K to 5000 K, then f = 5
If temp > 5000 K f = 7 [ 3 translational.+ 2 rotational + 2 vibrational ]
Maxwell’s Law Of Equpartition Of Energy
Energy associated with each degree of freedom = \(\frac{1}{2} \mathrm{kT}\). If the degree of freedom of a molecule is f, then the total kinetic energy of that molecule U = \(\frac{1}{2} \mathrm{fkT}\)
Internal Energy
The internal energy of a system is the sum of the kinetic and potential energies of the molecules of the system. It is denoted by U. Internal energy (U) of the system is the function of its absolute temperature (T) and its volume (V). i.e. U = f (T, V)
- In the case of an ideal gas, the intermolecular force is zero. Hence its potential energy is also zero.
- In this case, the internal energy is only due to kinetic energy, which depends on the absolute temperature of the gas. i.e. U = f (T).
For an ideal gas internal energy U = \(\frac{f}{2} n R T\)
Question 1. A light container having a diatomic gas enclosed within is moving with velocity v. The Mass of the gas is M and the number of moles is n.
- What is the kinetic energy of gas w.r.t. center of mass of the system?
- What is K.E. of gas w.r.t. ground?
Answer:
1. K.E. = \(\frac{5}{2} n R T\)

2. Kinetic energy of gas w.r.t. ground = Kinetic energy of center of mass w.r.t. ground + Kinetic energy of gas w.r.t. center of mass.
K.E. = \(\frac{1}{2} M V^2+\frac{5}{2} n R T\)
Question 2. Two nonconducting containers having volumes V1 and V2 contain monoatomic and diatomic gases respectively. They are connected as shown in the figure. Pressure and temperature in the two containers are P1, T1, and P2, T2 respectively. Initially stop cock is closed, if the stop cock is opened find the final pressure and temperature.

Answer:
⇒ \(n_1=\frac{P_1 V_1}{R T_1}\)
⇒ \(\mathrm{n}_2=\frac{\mathrm{P}_2 \mathrm{~V}_2}{R \mathrm{RT}_2}\)
n = n1+ n2(number of moles are conserved)
Finally, the pressure in both parts and the temperature of both gases will become equal.
⇒ \(\frac{P\left(V_1+V_2\right)}{R T}=\frac{P_1 V_1}{R T_1}+\frac{P_2 V_2}{R T_2}\)
From energy conservation
⇒ \(\frac{3}{2} n_1 R T_1+\frac{5}{2} n_2 R T_2\)
⇒ \(\frac{3}{2} n_1 R T+\frac{5}{2} n_2 R T\)
T = \(\frac{\left(3 P_1 V_1+5 P_2 V_2\right) T_1 T_2}{3 P_1 V_1 T_2+5 P_2 V_2 T_1}\)
P = \(\left(\frac{3 P_1 V_1+5 P_2 V_2}{3 P_1 V_1 T_2+5 P_2 V_2 T_1}\right)\left(\frac{P_1 V_1 T_2+P_2 V_2 T_2}{V_1+V_2}\right)\)
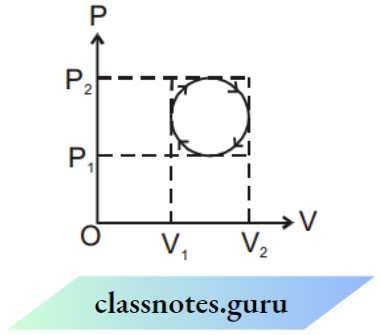
Indicator Diagram
A graph representing the variation of pressure or variation of temperature or variation of volume with each other is called or indicator diagram.

- Every point of the Indicator diagram represents a unique state (P, V, T) of gases.
- Every curve on the Indicator diagram represents a unique process.

Thermodynamics
Thermodynamics is mainly the study of the exchange of heat energy between bodies and the conversion of the same into mechanical energy and vice versa.
Thermodynamic System
The collection of an extremely large number of atoms or molecules confined within certain boundaries such that it has a certain value of pressure (P), volume (V), and temperature (T) is called a thermodynamic system.
Anything outside the thermodynamic system to which energy or matter is exchanged is called its surroundings. Taking into consideration the interaction between a system and its surroundings thermodynamic system is divided into three classes :
- Open system: A system is said to be an open system if it can exchange both energy and matter with its surroundings.
- Closed system: A system is said to be a closed system if it can exchange only energy (not matter with its surroundings).
- Isolated system: A system is said to be isolated if it can neither exchange nor matter with its surroundings.
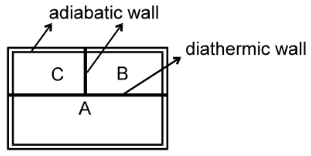
Zeroth Law Of Thermodynamics
If two systems (B and C) are separately in thermal equilibrium with a third one (A), then they are in thermal equilibrium with each other.
Class 11 NEET Physics Kinetic Theory of Gases and Thermodynamics Study Notes
Equation Of State ( For Ideal Gases)
The relation between the thermodynamic variables (P, V, T) of the system is called an equation of state. The equation of state for an ideal gas of n moles is given by PV = nRT,
Work Done By A Gas
Let P and V be the pressure and volume of the gas. If A is the area of the piston, then the force exerted by a gas on the piston is, F = P × A.
Let the piston move through a small distance dx during the expansion of the gas. Work done for a small displacement dx is dW = F dx = PA dx
Since A dx = dV, the increase in the volume of the gas is dV
⇒ dW = P dV
mg
or \(W=\int d W=\int P d V\)
The area enclosed under the P-V curve gives work done during the process.
Different Types Of Processes
1. Isothermal Process:
T = constant [Boyle’s law applicable] PV = constant
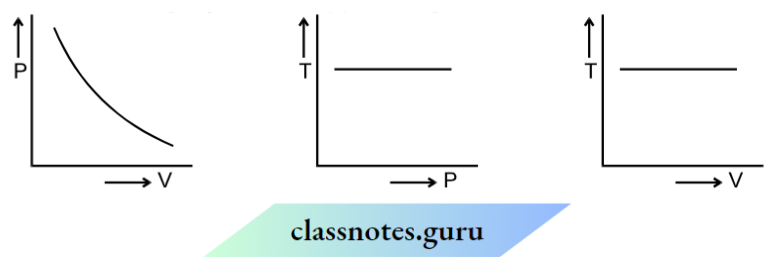
There is an exchange of heat between the system and its surroundings. The system should be compressed or expanded
very slowly so that there is sufficient time for the exchange of heat to keep the temperature constant.
The slope of the P−V curve in the isothermal process:
PV = constant = C
⇒ \(\frac{d P}{d V}=-\frac{P}{V}\)
Work done in the isothermal process:
W = \(n R T \quad \ell n \frac{V_f}{V_i}\)
[If vf > vi then W is positive
If vf < vi then W is negative]
W = \(\left[2.303 n R T \log _{10} \frac{V_f}{V_i}\right]\)

Internal energy in the isothermal process:
U = f (T) ⇒ ΔU = 0
2. Iso-Choric Process (Isometric Process):
V = constant
⇒ change in volume is zero
⇒ \(\frac{P}{T}\) is constant
⇒ \(\frac{P}{T}\)= const.(Galussac-law)
Work done in the isochoric process:
Since the change in volume is zero therefore dW = p dV = 0
Indicator diagram of the isochoric process:

Change in internal energy in isochoric process: ΔU = \(n \frac{f}{2} R \Delta T\)
Heat given in isochoric process: ΔQ = ΔU = \(n \frac{f}{2} R \Delta T\)
3. Isobaric Process: Pressure remains constant in the isobaric process
∴ P = constant
⇒ \(\frac{\mathrm{V}}{\mathrm{T}}\) = constant
Indicator diagram of the isobaric process:

Work done in the isobaric process:
ΔW = P ΔV = P (Vfinal – Vinitial) = nR (Tfinal – Tinitial)
Change in internal energy in the isobaric process: ΔU = n CVΔT
Heat given in the isobaric process:
ΔQ = ΔU + ΔW
ΔQ = \(n \frac{f}{2} R \Delta T+P\left[V_f-V_i\right]\)
⇒ \(n \frac{f}{2} R \Delta T+n R \Delta T\)
The above expression gives the idea that to increase the temperature by ΔT in the isobaric process heat required is more than in the isochoric process.
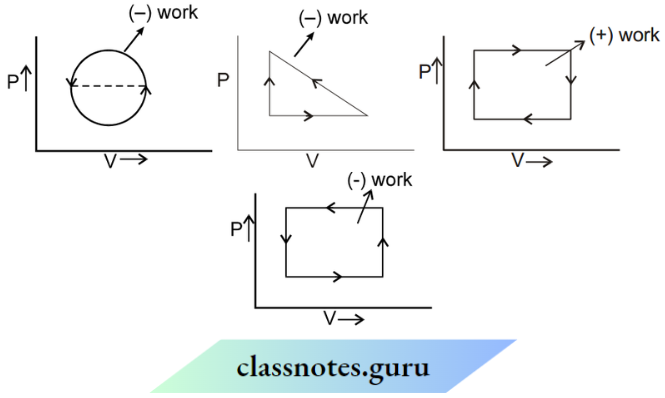
4. Cyclic Process: In the cyclic process initial and final states are the same therefore initial state = final state
Work done = Area enclosed under P-V diagram.
Change in internal Energy ΔU = 0
ΔQ = ΔU + ΔW
∴ ΔQ = ΔW
If the process on the P-V curve is clockwise, then the net work done is (+ve) and vice-versa. The graphs shown below explain when work is positive and when it is negative
Question 1. The cylinder shown in the figure has conducting walls and the temperature of the surroundings is T, the position is initially in equilibrium, and the cylinder contains n moles of a gas. Now the piston is displaced slowly by an external agent to make the volume double the initial. Find work done by an external agent in terms of n, R, T

Answer:
1st Method:
Work done by external agents is positive because Fext and displacement are in the same direction. Since walls are conducting therefore temperature remains constant.

Applying the equilibrium condition when the pressure of the gas is P
⇒ \(P A+F_{\text {ext }}=P_{a t m} A\)
⇒ \(F_{\text {ext }}=P_{a t m} A-P A\)
⇒ \(W_{e x t}=\int_0^d F_{e x t} d x=\int_0^d P_{a t m} A d x-\int_0^d P A d x \)
⇒ \(P_{a t m} A \int_0^d d x-\int_V^{2 V} \frac{n R T}{V} d V=P_{a t m} A d-n R T \ln 2\)
⇒ \(P_{a t m} \cdot V_0^2-n R T \ln 2=n R T(1-\ln 2)\)
2nd Method
Applying the work-energy theorem on the piston
Δk = 0
Wall = Δk
Wgas+ Watm + Wext = 0
⇒ \(n R T \ln \frac{V_f}{V_i}-n R T+W_{\text {ext }}=0\)
Wext = nRT (1 – ln2)
Question 2. Find out the work done in the given graph. Also, draw the corresponding T-V curve and P-T curve.

Answer:
Since in P-V curves area under the cycle is equal to work done therefore work done by the gas is equal to P0 V0. Lines A B and CD are isochoric lines, and lines BC and DA are isobaric lines.
∴ The T-V curve and P-T curve are drawn as shown.

Question 3. The t-V curve of the cyclic process is shown below, number of moles of the gas is n to find the total work done during the cycle.
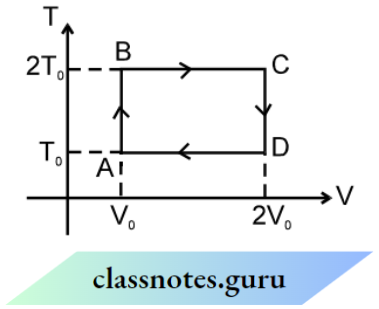
Answer: Since path AB and CD are isochoric therefore work done is zero during path AB and CD. Process BC and DA are isothermal, therefore
⇒ \(W_{B C} =n R 2 T_0 \ln \frac{V_C}{V_B}=2 n R T_0 \ln 2\)
⇒ \(W_{D A} =n R T_0 \ln \frac{V_A}{V_D}=-n R T_0 \ln 2\)
Total work done = \(W_{B C}+W_{D A}=2 n R T_0 \ln 2-n R T_0 \ln 2\)
⇒ \(n R T_0 \ln 2\)
Question 4. The P-T curve of a cyclic process is shown. Find out the work done by the gas in the given process if several moles of the gas are n.

Answer: Since path AB and CD are isochoric therefore work done during AB and CD is zero. Path BC and DA are isobaric.
Hence WBC = nRΔT = nR(T3– T2)
WDA = nR(T1– T4)
Total work done = WBC + WDA = nR(T1+ T3–T4– T2)
Question 5. Consider the cyclic process ABCA on a sample of 2.0 mol of an ideal gas as shown in the figure. The temperatures of the gas at A and B are 300 K and 500 K respectively. A total of 1200 J heat is withdrawn from the sample in the process. Find the work done by the gas in part BC. Take R = 8.3J/mol–K.
Answer:
The change in internal energy during the cyclic process is zero. Hence, the heat supplied to the gas is equal to the work done by it. Hence,
WAB + WBC + WCA = –1200 J. …….(1)
The work done during the process AB is

WAB = PA(VB– VA)
= nR(TB– TA)
= (2.0 mol) (8.3 J/mol–K) (200 K)
= 3320 J
The work done by the gas during the process CA is zero as the volume remains constant. From (1),
3320 J + WBC = –1200 J
or WBC = –4520 J.
= –4520 J.
First Law Of Thermodynamics
The first law of thermodynamics is the law of conservation of energy. It states that if a system absorbs heat dQ and as a result the internal energy of the system changes by dU and the system does a work dW, then dQ = dU + dW.
But, do = P dV dQ = dU + P dV
which is the mathematical statement of the first law of thermodynamics.
Heat gained by a system, work done by a system, and an increase in internal energy is taken as positive.
Heat lost by a system, work done on a system, and a decrease in internal energy are taken as negative.
Question 1. 1 gm water at 100ºC is heated to convert into steam at 100ºC at 1 atm. Find out the change in the internal energy of water. It is given that a volume of 1 gm water at 100ºC = 1 cc. volume of 1 gm steam at 100ºC = 1671 cc. Latent heat of vaporization = 540 cal/g. (Mechanical equivalent of heat J = 4.2J/cal.)
Answer:
From the first law of thermodynamic ΔQ = Δu + Δw
ΔQ = mL = 1 × 540 cal. = 540 cal.
ΔW = PΔV = \(\frac{10^5(1671-1) \times 10^{-6}}{4.2}\)
⇒ \(\frac{\left.10^5 \times 1670\right) \times 10^{-6}}{4.2}\) = 40 cal.
Δu = 540 – 40 = 500 cal.
Question 2. Two moles of diatomic gas at 300 K are kept in a non-conducting container enclosed by a piston. Gas is now compressed to increase the temperature from 300 K to 400 K. Find work done by the gas

Answer:
ΔQ = Δu + Δw
Since the container is non-conducting therefore
ΔQ = 0 = Δu + Δw
⇒ ΔW = – Δu = \(-\Delta u=-n \frac{f}{2} R \Delta T\)
⇒ \(-2 \times \frac{5}{2} R(400-300)\)
= –5 × 8.314 × 100 J
= – 5 × 831.4 J
= –4157 J
NEET Physics Chapter 9 Kinetic Theory of Gases and Thermodynamics: Key Concepts
Question 3. A sample of an ideal gas is taken through the cyclic process of abaca (figure. It absorbs 50 J of heat during part ab, no heat during bc, and rejects 70 J of heat during ca. 40 J of work done on the gas during part bc.
- Find the internal energy of the gas at b and c if it is 1500 J at a.
- Calculate the work done by the gas during the part ca.

Answer:
1. In part ab the volume remains constant. Thus, the work done by the gas is zero. The heat absorbed by the gas is 50 J. The increase in internal energy from a to b is
ΔU = ΔQ = 50J.
As the internal energy is 1500 J at a, it will be 1550 J at b. In part bc, the work done by the gas is ΔW = –40J and no heat is given to the system. The increase in internal energy from b to c is
ΔU = –ΔW = 40 J.
As the internal energy is 1550 J at b, it will be 1590 J at c.
2. The change in internal energy from c to a is
ΔU = 1500 J – 1590 J = – 90 J.
The heat given to the system is ΔQ = – 70J.
Using ΔQ = ΔU + ΔW,
ΔWca= ΔQ – ΔU
= – 70 J + 90 J
= 20 J.
Question 4. The internal energy of a monatomic ideal gas is 1.5 nRT. One mole of helium is kept in a cylinder of cross-section 8.5 cm2. The cylinder is closed by a light frictionless piston. The gas is heated slowly in a process during which a total of 42 J heat is given to the gas. If the temperature rises through 2ºC, find the distance moved by the piston. Atmospheric pressure = 100 kPa.
Answer:
The change in internal energy of the gas is
ΔU = 1.5 nR (ΔT)
= 1.5 (1 mol) (8.3 J/mol-K) (2K)
= 24.9 J.
The heat is given to the gas = 42 J
The work done by the gas is
ΔW = ΔQ – ΔU
= 42 J – 24.9 J = 17.1 J.
If the distance moved by the piston is x, the work done is
ΔW = (100 kPa) (8.5 cm2) x.
Thus, (105 N/m2) (8.5 × 10-4 m2) x = 17.1 J
or, x = 0.2m = 20 cm.
Question 5. A sample of ideal gas (f =5) is heated at constant pressure. If an amount of 140 J of heat is supplied to the gas, find
- The change in internal energy of the gas
- The work done by the gas.
Answer:
Suppose the sample contains n moles. Also, suppose the volume changes from V1 to V2 and the temperature changes from T1 to T2.
The heat supplied is
ΔQ = ΔU + PΔV = ΔU + nRΔT = \(\Delta U+\frac{2 \Delta U}{f}\)
1. The change in internal energy is
ΔU = \(n \frac{f}{2} R\left(T_2-T_1\right)=n \frac{f}{2} R\left(T_2-T_1\right)\)
⇒ \(\frac{\mathrm{f}}{2+\mathrm{f}} \Delta Q=\frac{140 \mathrm{~J}}{1.4}\)
= 100 J.
2. The work done by the gas is
ΔW = ΔQ – ΔU
= 140 J – 100 J
= 40 J.
Efficiency Of A Cycle (η):
⇒ \(\eta=\frac{\text { total Mechanical work done by the gas in the whole process }}{\text { Heat absorbed by the gas (only }+ \text { ve) }}\)
⇒ \(=\frac{\text { area under the cycle in } \mathrm{P}-\mathrm{V} \text { curve }}{\text { Heat injected into the system }}\)
⇒ \(\eta=\left(1-\frac{Q_2}{Q_1}\right)\) for Heat Engine,
⇒ \(\eta=\left(1-\frac{T_2}{T_1}\right)\) for Carnot cycle
Question 6. n moles of a diatomic gas have undergone a cyclic process ABC as shown in the figure. The temperature at a is T0. Find

- Volume at C?
- Maximum temperature?
- Total heat given to gas?
- Is heat rejected by the gas, if yes how much heat is rejected?
- Find out the efficiency
Answer:
1. Since triangles OA V0 and OC V are similar therefore
⇒ \(\frac{2 P_0}{V}=\frac{P_0}{V_0}\)
⇒ \(V=2 V_0\)
2. Since process AB is isochoric hence
⇒ \(\frac{P_A}{T_A}=\frac{P_B}{T_B}\)
⇒ \(T_B=2 T_0\)
Since process BC is isobaric therefore \(\frac{T_B}{V_B}=\frac{T_C}{V_C}\)
⇒ \(T_{\mathrm{C}}=2 \mathrm{~T}_{\mathrm{B}}=4 \mathrm{~T}_0\)
3. Since the process is cyclic therefore
∴ ΔQ = ΔW = area under the cycle = \(\frac{1}{2} P_0 V_0\)
4. Since Δu and ΔW both are negative in process CA
∴ ΔQ is negative in process CA and heat is rejected in process CA
ΔQCA = ΔwCA + ΔuCA
⇒ \(-\frac{1}{2}\left[P_0+2 P_0\right] V_0-\frac{5}{2} n R\left(T_c-T_a\right)\)
⇒ \(-\frac{1}{2}\left[P_0+2 P_0\right] V_0-\frac{5}{2} n R\left(\frac{4 P_0 V_0}{n R}-\frac{P_0 V_0}{n R}\right)\)
= –9P0V0
= Heat injected.
4. η = efficiency of the cycle = \(=\frac{\text { work done by the gas }}{\text { heat injected }}\)
⇒ \(\eta=\frac{P_0 V_0 / 2}{Q_{\text {injected }}} \times 100\)
ΔQinj = ΔQAB + ΔQBC
⇒ \(\left[\frac{5}{2} n R\left(2 T_0-T_0\right)\right]+\left[\frac{5}{2} n R\left(2 T_0\right)+2 P_0\left(2 V_0-V_0\right)\right]\)
⇒ \(\frac{19}{2} P_0 V_0\)
⇒ \(\eta=\frac{100}{19} \%\)
Specific Heat
The specific heat capacity of a substance is defined as the heat supplied per unit mass of the substance per unit rise in the temperature. If an amount ΔQ of heat is given to a mass m of the substance and its temperature rises by ΔT, the specific heat capacity s is given by the equation
⇒ \(s=\frac{\Delta Q}{m \Delta T}\)
The molar heat capacities of a gas are defined as the heat given per mole of the gas per unit rise in the temperature. The molar heat capacity at constant volume, denoted by Cv, is :
⇒ \(C_v=\left(\frac{\Delta Q}{n \Delta T}\right)_{\text {constant volume }}\)
⇒ \(\frac{f}{2} R\)
and the molar heat capacity at constant pressure, denoted by CP is,
⇒ \(C_{\mathrm{P}}=\left(\frac{\Delta Q}{\mathrm{n} \Delta T}\right)_{\text {constant volume }}\)
⇒ \(\left(\frac{f}{2}+1\right) R\)
- where n is the amount of the gas in several moles and f is a degree of freedom. Quite often, the term specific heat capacity or specific heat is used for molar heat capacity.
- It is advised that the unit be carefully noted to determine the actual meaning. The unit of specific heat capacity is J/kg-K whereas that of molar heat capacity is J/mol–K.
Molar Heat Capacity Of Ideal Gas In Terms Of R:
1. For a monoatomic gas f = 3
⇒ \(C_v=\frac{3}{2} R\)
⇒ \(C_p=\frac{5}{2} R\)
⇒ \(\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}=\gamma=\frac{5}{3}\)
= 1.67
2. For a diatomic gas f = 5
⇒ \(\mathrm{C}_{\mathrm{V}}=\frac{5}{2} \mathrm{R},\)
⇒ \(\mathrm{C}_{\mathrm{P}}=\frac{7}{2} \mathrm{R}\)
⇒ \(\gamma=\frac{C_P}{C_V}\)
= 1.4
3. For a Triatomic gas f = 6
⇒ \(C_V=3 R, C_P=4 R\)
⇒ \(\gamma=\frac{C_P}{C_V}=\frac{4}{3}\) = 1.33 [Note for CO2; f = 5, it is linear]
In general, if f is the degree of freedom of a molecule, then,
⇒ \(C_V=\frac{f}{2} R, \quad C_P=\left(\frac{f}{2}+1\right) R\)
⇒ \(\gamma=\frac{C_P}{C_V}=\left[1+\frac{2}{f}\right]\)
Question 1. In a thermodynamic process, the pressure of a certain mass of gas is changed in such a way that 20 Joule heat is released from it and 8 Joule work is done on the gas. If the initial internal energy of the system is 30 joule then the final internal energy will be –
Answer:
dQ = dU + dW ⇒ dQ = Ufinal– Uinitial + dW
Ufinal = dQ – dW + Uinitial or Ufinal = –20 + 8 + 30 of Ufinal = 18 Joule
Question 2. A gas is contained in a vessel fitted with a movable piston. The container is placed on a hot stove. A total of 100 cal of heat is given to the gas and the gas does 40 J of work in the expansion resulting from heating. Calculate the increase in internal energy in the process.
Answer:
Heat given to the gas is ΔQ = 100 cal = 418 J.
Work done by the gas is ΔW = 40 J
The increase in internal energy is
ΔU = ΔQ – ΔW
= 418J – 40 J = 378 J
Question 3. A gas is compressed from volume 10 m3 to 4 m3 at a constant pressure of 50N/m2. Gas is given 100 J energy by heating then its internal energy.
Answer:
P = 50 N/m2
dV = 10 – 4 = 6 m3
δW = PdV = 6 × 50 = 300 J (Volume is decreasing, δQ = 100 J)
W = – 300 J
δQ = δW + dU
100 + 300 = dU
dU = increased by 400 J
Question 4. Two moles of a diatomic gas at 300 K are enclosed in a cylinder as shown in the figure. The piston is light. Find out the heat given if the gas is slowly heated to 400 K in the following three cases.

- The piston is free to move
- If the piston does not move
- If the piston is heavy and movable.
Answer:
1. Since pressure is constant
∴ ΔQ = nCP ΔT = 2 × \(\frac{7}{2}\) 2× R × (400 – 300) = 700 R
2. Since volume is constant
∴ ΔW = 0 and ΔQ = Δu (from first law)
ΔQ = Δu = nCvΔT = 2× \(\frac{5}{2}\) × R × (400 – 300) = 500 R
3. Since pressure is constant
∴ ΔQ = nCP ΔT = 2 × \(\frac{7}{2}\) × R × (400 – 300) = 700 R
Question 5. The p-V curve of a diatomic gas is shown in the figure. Find the total heat given to the gas in the process AB and BC
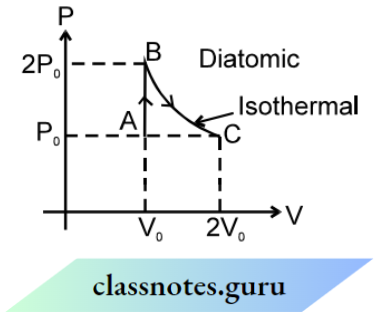
Answer:
From the first law of thermodynamics
ΔQABC = ΔuABC + ΔWABC
⇒ \(\Delta W_{A B C}=\Delta W_{A B}+\Delta W_{B C}=0+n R T_B \ln \frac{V_C}{V_B}=n R T_B \ln \frac{2 V_0}{V_0}\)
⇒ \(n R T_B \ln 2=2 P_0 V_0 \ln 2\)
Δu = \(n C_v \Delta T=\frac{5}{2}\left(2 P_0 V_0-P_0 V_0\right)\)
⇒ ΔQABC = \(\frac{5}{2}\) 2P0V0+ 2P0V0ln 2.
Question 6. Calculate the value of the mechanical equivalent of heat from the following data. The specific heat capacity of air at constant volume = 170 cal/kg-K, γ=CP/Cv= 1.4, and the density of air at STP is 1.29 kg/m3. Gas consant R = 8.3 J/mol-K.
Answer:
Using pV = nRT, the volume of 1 mole of air at STP is
V = \(\frac{\mathrm{nRT}}{\mathrm{p}}=\frac{(1 \mathrm{~mol}) \times(8.3 \mathrm{~J} / \mathrm{mol}-\mathrm{K}) \times(273 \mathrm{~K})}{1.0 \times 10^5 \mathrm{~N} / \mathrm{m}^2}\)
= 0.0224m3
The mass of 1 mole is, therefore,
(1.29 kg/m3) × (0.0224 m3) = 0.029 kg.
The number of moles in 1 kg is \(\frac{1}{0.029}\). The molar heat capacity at constant volume is
⇒ \(C_v=\frac{170 \mathrm{cal}}{(1 / 0.029) \mathrm{mol}-\mathrm{K}}\)= 4.93 cal/mol-K.
Hence, CP = γCv= 1.4 × 4.93 cal/mol-K
or, CP – Cv= 0.4 × 4.93 cal/mol-K
= 1.97 cal/mol-K.
Also, CP– Cv= R = 8.3 J/mol-K.
Thus, 8.3 J = 1.97 cal.
The mechanical equivalent of heat is
⇒ \(\frac{8.3 \mathrm{~J}}{1.97 \mathrm{cal}}\) =4.2 J/cal.
Average Molar Specific Heat of Metals:
[Dulong and Petit law]
At room temperature average molar specific heat of all metals are same and is nearly equal to 3R ( 6 cal. mol-1 K-1).
[Note: Temp. above which the metals have constant CV is called Debye temp.]

Mayer’s Equation: CP− CV= R (for ideal gases only)
Adiabatic Process:
When no heat is supplied or extracted from the system the process is called adiabatic. The process is sudden so there is no time for the exchange of heat. If the walls of a container are thermally insulated no heat can cross the boundary of the system and the process is adibatic.

The equation of the adiabatic process is given by
PVγ = constant [Poission Law]
Tγ P1-γ = constant
T Vγ-1 = constant
The slope of P−V−curve in the adiabatic process:
Since PVγ is a constant
∴ \(\frac{\mathrm{dP}}{\mathrm{dV}}=-\gamma\left(\frac{\mathrm{P}}{\mathrm{V}}\right)\)

The slope of P−T−curve in the adiabatic process: Since Tγ P1-γ is a constant
∴ \(\frac{d P}{d T}=-\frac{\gamma}{(1-\gamma)} \frac{P}{T}=\frac{(\gamma)}{(\gamma-1)} \frac{P}{T}\)

Slope of T−V−curve:
⇒ \(\frac{d V}{d T}=-\frac{1}{(\gamma-1)} \frac{V}{T}\)

Work Done In Adiabatic Process:
\(\Delta W=-\Delta U=n C_v\left(T_i-T_f\right)=\frac{P_i V_i-P_f V_f}{(\gamma-1)}=\frac{n R\left(T_i-T_f\right)}{\gamma-1}\)
work done by the system is (+ve), if Ti> Tf (hence expansion)
work done on the system is (−ve) if Ti< Tf (hence compression)
Question 7. A quantity of air is kept in a container having walls that are slightly conducting. The initial temperature and volume are 27ºC (equal to the temperature of the surroundings) and 800cm3 respectively. Find the rise in the temperature if the gas is compressed to 200cm3
- In a short time
- In a long time. Take γ = 1.4.
Answer:
1. When the gas is compressed in a short time, the process is adiabatic. Thus,
⇒ \(\mathrm{T}_2 \mathrm{~V}_2^{\gamma-1}=\mathrm{T}_1 \mathrm{~V}_1^{\gamma-1}\)
or \(\mathrm{T}_2=\mathrm{T}_1\left(\frac{\mathrm{V}_1}{\mathrm{~V}_2}\right)^{\gamma-1}\)
⇒ \((300 \mathrm{~K}) \times\left[\frac{800}{200}\right]^{0.4}\)
= 522K
Rise in temperature = T2– T1= 222 K.
2. When the gas is compressed for a long time, the process is isothermal. Thus, the temperature remains equal to the temperature of the surroundings which is 27ºC. The rise in temperature = 0.
NEET Physics Chapter 9 Notes: Kinetic Theory of Gases and Thermodynamics
Question 8. A monoatomic gas is enclosed in a nonconducting cylinder having a piston that can move freely. Suddenly gas is compressed to 1/8 of its initial volume. Find the final pressure and temperature if the initial pressure and temperature are P0 and T0 respectively.

Answer:
Since the process is adiabatic therefore
⇒ \(P_0 V^{\frac{5}{3}}=P_{\text {final }}\left(\frac{V}{8}\right)^{5 / 3}\)
⇒ \(\gamma=\frac{C_P}{C_V}=\frac{5 R}{2} / \frac{3 R}{2}=\frac{5}{3}\)
⇒ \(P_{\text {final }}=32 P_0\)
Since the process is adiabatic therefore
⇒ \(\mathrm{T}_1 \mathrm{~V}_1^{\gamma-1}=\mathrm{T}_2 \mathrm{~V}_2^{\gamma-1}\)
⇒ \(\mathrm{T}_0 \mathrm{~V}_0^{2 / 3}=\mathrm{T}_{\text {frall }}\left(\frac{\mathrm{V}_0}{8}\right)^{2 / 3}\)
⇒ \(\mathrm{T}=4 \mathrm{~T}_0\)
Question 9. A cylindrical container having nonconducting walls is partitioned into two equal parts such that the volume of each part is equal to V0. A movable nonconducting piston is kept between the two parts. The gas on the left is slowly heated so that the gas on the right is compressed upto volume \(\frac{V_0}{8}\). Find pressure and temperature on both sides if the initial pressure and temperature were P0 and T0 respectively. Also, find heat given by the heater to the gas. (number of moles in each part is n)

Answer:
Since the process on the right is adiabatic therefore PVγ = constant
⇒ P0 Vγ0 = Pfinal (V0/ 8)γ ⇒ Pfinal = 32
P0 T0 Vγ-10 = Tfinal (V0/8)γ-1 ⇒ Tfinal = 4T0
Let the volume of the left part be V1
⇒ \(2 V_0=V_1+\frac{V_0}{8}\)
⇒ \(V_1=\frac{15 V_0}{8}\)
Since several moles on the left parts remain constant therefore for the left part
⇒ \(\frac{\mathrm{PV}}{\mathrm{T}}\) = constant.
The final pressure on both sides will be the same
⇒ \(\frac{P_0 V_0}{T_0}=\frac{P_{\text {final }} V_1}{T_{\text {final }}}\)
⇒ \(\mathrm{T}_{\text {final }}=60 \mathrm{~T}_0\)
⇒ \(\Delta Q=\Delta u+\Delta w\)
⇒ \(\Delta Q=n \frac{5 R}{2}\left(60 T_0-T_0\right)+n \frac{3 R}{2}\left(4 T_0-T_0\right)\)
⇒ \(\Delta Q=\frac{5 n R}{2} \times 59 T_0+\frac{3 n R}{2} \times 3 T_0\)
Free Expansion
If a system, say a gas expands in such a way that no heat enters or leaves the system and also no work is done by or on the system, then the expansion is called the “free expansion”.
ΔQ = 0 , ΔU = 0 and ΔW = 0. Temperature in the free expansion remains constant.
Question 1. A nonconducting cylinder having volume 2V0 is partitioned by a fixed nonconducting wall in two equal parts. Partition is attached with a valve. The right side of the partition is a vacuum and the left part is filled with a gas having pressure and temperature P0 and T0 respectively. If the valve is opened the final pressure and temperature of the two parts are.
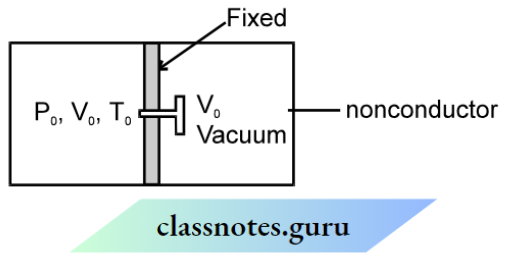
Answer: From the first law of thermodynamics ΔQ = Δu + ΔW
Since gas expands freely, therefore, ΔW = 0 since no heat is given to gas ΔQ = 0
⇒ Δu = 0 and temperature remains constant.
Tfinal = T0
Since the process is isothermal therefore P0× V0= Pfinal × 2V0
⇒ Pfinal = P0/2
Reversible And Irreversible Process
A process is said to be reversible when the various stages of an operation in which it is subjected can be traversed back in the opposite direction in such a way that the substance passes through the same conditions at every step in the reverse process as in the direct process.
A process in which any one of the conditions stated for the reversible process is not fulfilled is called an irreversible process.
Comparison Of Slopes Of Iso-Thermal And Adiabatic Curve

In compression up to the same final volume: | Wadia |> | Wisothermal |
In Expansion up to the same final volume: Wisothermal > Wadia
Limitations Of Ist Law Of Thermodynamics:
The first law of thermodynamics tells us that heat and mechanical work are interconvertible. However, this law fails to explain the following points:
- It does not tell us about the direction of the transfer of heat.
- It does not tell us about the conditions under which heat energy is converted into work.
- It does not tell us whether some process is possible or not.
Mixture Of Non-Reacting Gases:
1. Molecular weight = \(\frac{\mathrm{n}_1 \mathrm{M}_1+\mathrm{n}_2 \mathrm{M}_2}{\mathrm{n}_1+\mathrm{n}_2}\)
M1 and M2 are molar masses.
2. Specific heat CV = \(\frac{\mathrm{n}_1 \mathrm{C}_{\mathrm{V}_1}+\mathrm{n}_2 \mathrm{C}_{\mathrm{V}_2}}{\mathrm{n}_1+\mathrm{n}_2}\)
⇒ \(C_p=\frac{n_1 C_{P_1}+n_2 C_{P_2}}{n_1+n_2}\)
3. For mixture, γ = \(\gamma=\frac{\mathrm{C}_{{mix}}}{\mathrm{C}_{\mathrm{v}_{{mix}}}}\)
⇒ \(\frac{n_1 C_{p_1}+n_2 C_{p_2}+\ldots \ldots}{n_1 C_{v_1}+n_2 C_{v_2}+\ldots \ldots \ldots}\)
Question 1. 5 gm air is heated from 4ºC to 6ºC. If the specific heat of air at constant volume is 0.172 cal/gm/ºC, then the increase in the internal energy of air will be –
Answer:
dU = mCvdT
dU = 5 × 0.172 × 2
dU = 1.72 calorie
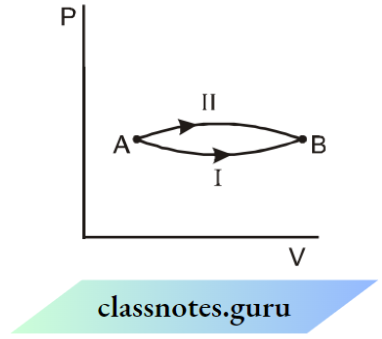
Question 2. In the following indicator diagram, the net amount of work done will be –
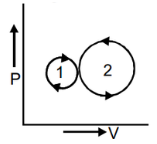
Answer:
The cyclic process 1 is clockwise and the process 2 is anti clockwise. Therefore W1 will be positive and W2 will be negative area 2 > area 1, Hence the network will be negative.
Kinetic Theory of Gases NEET Notes for Class 11
Question 3. Two gram-moles of gas, which are kept at a constant temperature of 0ºC, are compressed from 4 liter to 1 liter. The work done will be
Answer:
W = 2.303 μ RT \(\log _{10} \frac{V_2}{V_1}\)
W = 2.303 × 2 × 8.4 × 273 \(\log _{10} \frac{1}{4}\)
W = 2.303 × 2 × 8.7 × 273 × (log10 – log410)
∵ log410 = 0.6021
∴ W = –6359 Joule
Question 4. Air is filled in a motor car tube at 27ºC temperature and 2-atmosphere pressure. If the tube suddenly bursts then the final temperature will be \(\left[\left(\frac{1}{2}\right)^{2 / 7}=0.82\right]\)
Answer:
⇒ \(\mathrm{T}_2=\mathrm{T}_1\left(\frac{\mathrm{P}_2}{\mathrm{P}_1}\right)^{\frac{\gamma-1}{\gamma}}\)
⇒ \(\mathrm{T}_2=300\left(\frac{1}{2}\right)^{\frac{0.4}{1.4}}\)
⇒ \(300\left(\frac{1}{2}\right)^{2 / 7}\)
= 300×8.2
T2 = 246K
Question 5. One liter of air at NTP is suddenly compressed to 1 c.c. the final pressure will be.
Answer:
⇒ \(P_2=P_1\left(\frac{V_1}{V_2}\right)^\gamma\)
⇒ \(P_2=10^5\left(10^3\right)^{5 / 3}=10^5 \times 10^5\)
⇒ \(P_2=10^{10} \text { Pascal }\)
Question 6. In the following fig. the work done by the system in the closed path ABCA is Answer: Work done in closed path ABCA
WABCA = Area of ΔABC = \(\frac{1}{2}\) AB × BC
WABCA = –\(\frac{1}{2}\)(P2– P1) (V2– V1)
Question 7. According to the fig. if one mole of an ideal gas is in a cyclic process the work done by the gas in the process will be
Answer:
Work done W = area of PV curve
⇒ \(\frac{1}{2}\)[3P0– P0][2V0– V0]
= P0V0
Question 8. In the above question, heat given by the gas is
Answer:
δQ = μCpdT, μ = 1, dT = TA– TC, and for monoatomic ideal gas CP= 5/2 R
∴ (δQ)CA = \(\frac{5}{2}\) R [TA– TC] = \(\frac{5}{2}\)[PAVA– PCVC]
But PA= P0, VA= V0, VC= 2V0, PC= P0
and \(\frac{PV}{T}\)
∴ (δQ)CA = \(\frac{5}{2}\)[P0V0– P02V0] = – \(\frac{5}{2}\)P0V0
Question 9. In the above question, the heat taken by gas in the path AB will be Answered:
(δQ)AB = μCVdT (the process is on constant volume)
CV= \(\frac{3}{2}\)R, μ = 1
⇒ \((\delta Q)_{A B}=\frac{3}{2} R\left[T_B-T_A\right]\)
⇒ \(\frac{3}{2}\left[3 P_0 V_0-P_0 V_0\right]\)
⇒ \(3 \mathrm{P}_0 \mathrm{~V}_0\)
Question 10. In the above question, the absorbed heat by gas in path BC will be Answered:
If the heat given for the complete process is δQ then
(δQ) = (δQ)AB + (δQ)BC + (δQ)CA
dU = 0 in a cyclic process, thus by the first law of thermodynamics
δQ = δW
∴ (δQ)AB + (δQ)BC + (δQ)AC = δW
(δQ)BC = δW – (δQ)AB – (δQ)BC
⇒ \(P_0 V_0+\frac{5}{2} P_0 V_0-3 P_0 V_0\)
⇒ \(\frac{P_0 V_0}{2}\)
Question 11. For a given cyclic process as shown in fig. the magnitude of absorbed energy for the system is
Answer:
In cyclic process
Q = W (∵ dU = 0)
Q = area of closed loop
Q = 102π Joule
Second Law Of Thermodynamics
This law gives the direction of heat flow.
According To Classius: It is impossible to make any such machine that can transfer heat from an object with low temperature to an object with high temperature without an external source.
According To Kelvin: It is impossible to obtain work continuously by cooling an object below the temperature of its surroundings.
Statement Of Kelvin-Planck: It is impossible to construct any such machine that works on a cyclic process and absorbs heat from a source, converts all that heat into work, and rejects no heat to sink.
Heat Engine:
The device, used to convert heat energy into mechanical energy, is called a heat engine.
- For the conversion of heat into work with the help of a heat engine, the following conditions have to be met. There should be a body at a higher temperature ‘T1’ from which heat is extracted. It is called the source.
- The body of the engine contains the working substance. There should be a body at a lower temperature ‘T2’ to which heat can be rejected. This is called the sink.
Working Of Heat Engine:
Schematic diagram of heat engine

Engine derives an amount ‘Q1’ of heat from the source.
A part of this heat is converted into work ‘W’.
The remaining heat ‘Q2’ is rejected to the sink.
Thus Q1= W + Q2
or the work done by the engine is given by W = Q1– Q2
Efficiency Of Heat Engine:
The efficiency of the heat engine (η) is defined as the fraction of total heat, supplied to the engine which is converted into work.
Mathematically
∴ \(\eta=\frac{W}{Q_1}\)
or \(\eta=\frac{Q_1-Q_2}{Q_1}=1-\frac{Q_2}{Q_1}\)
Carnot Engine And Carnot Cycle
Carnot Engine: The Carnot engine is an ideal heat engine. It consists of the following parts.
Schematic Diagram:

Source: It is a reservoir of heat energy with a conducting top maintained at a constant temperature T1K. The source is so big that extraction of any amount of heat from it does not change its temperature.
Body Of Heat Engine: It is a barrel having perfectly insulating walls and a conducting bottom. It is fitted with an air-tight piston capable of sliding within the barrel without friction. The barrel contains some quantity of an ideal gas.
Sink: It is a huge body at a lower temperature T2 having a perfectly conducting top. The size of the sink is so large that any amount of heat rejected to it does not increase its temperature.
Insulating Stand: It is a stand made up of perfectly insulating material such that the barrel when placed over it becomes thoroughly insulated from the surroundings.
Carnot Cycle: As the engine works, the working substance of the engine undergoes a cycle known as the Carnot cycle. The Carnot cycle consists of the following four strokes.
Graphical representation of the Carnot cycle:
First Stroke (Isothermal expansion): In this stroke, the barrel is placed over the source. The piston is gradually pushed back as the gas expands.
The fall of temperature, due to expansion, is compensated by the supply of heat from the source and consequently, temperature remains constant. The conditions of the gas change from A(P1, V1) to B(P2, V2). If W1 is the work done during this process, then heat Q1 derived from the source is given by
⇒ \(\mathrm{Q}_1=\mathrm{W}_1=\text { Area } \mathrm{ABGE}=\mathrm{RT} \log _e\left(\frac{\mathrm{V}_2}{\mathrm{~V}_1}\right)\)
Second Stroke (Adiabatic expansion):
The barrel is removed from the source and placed over the insulating stand. The piston is pushed back so that the gas expands adiabatically resulting in a fall of temperature from T1 to T2. The conditions of the gas change from B(P2, V2) to C(P3, V3). If W2 is the work done in this case then
⇒ \(\mathrm{W}_2=\text { Area } \mathrm{BCHG}=\frac{\mathrm{R}}{\gamma-1}\left(\mathrm{~T}_1-\mathrm{T}_2\right)\)
Third Stroke (isothermal compression): The barrel is placed over the sink. The piston is pushed down there by compressing the gas. The heat generated due to compression flows to the sink maintaining the temperature of the barrel constant.
The state of the gas changes from C(P3, V3) to D(P4, V4). If W3 is the work done in this process and Q2 is the heat rejected to the sink, then
⇒ \(\mathrm{Q}_2=\mathrm{W}_3=\text { Area CDFH }=\mathrm{RT}_2 \log _{\mathrm{e}}\left(\frac{\mathrm{V}_3}{\mathrm{~V}_4}\right)\)
Fourth Stroke (Adiabatic compression): The barrel is placed over the insulating stand. The piston is moved down thereby compressing the gas adiabatically till the temperature of the gas increases from T2 to T1.
The state of gas changes from D(P4, V4) to A(P1, V1). If W4 is the work done in this process, then
⇒ \(\mathrm{W}_4=\text { Area ADFE }=\frac{\mathrm{R}}{\gamma-1}\left(\mathrm{~T}_1-\mathrm{T}_2\right)\)
Heat Converted Into Work In Carnot Cycle:
During the four strokes, W1 and W2 are the work done by the gas, and W3 and W4 are the work done on the gas. Therefore the net, work performed by the engine
W = W1+ W2– W3– W4= Area ABGE + Area BCHG – Area CDFH – Area ADEF = Area ABCD
Thus net work done by the engine during one cycle is equal to the area enclosed by the indicator diagram of the cycle. Analytically
⇒ \(\mathrm{W}=\mathrm{R}\left(\mathrm{T}_1-\mathrm{T}_2\right) \log _e\left(\frac{\mathrm{V}_2}{\mathrm{~V}_1}\right)\)
Efficiency Of Carnot Engine:
The efficiency (η) of an engine is defined as the ratio of useful heat (heat converted into work) to the total heat supplied to the engine. Thus.
⇒ \(\eta=\frac{W}{Q_1}=\frac{Q_1-Q_2}{Q_1}\)
or \(\eta=\frac{R\left(T_1-T_2\right) \log _e\left(\frac{V_2}{V_1}\right)}{R T_1 \log _e\left(\frac{V_2}{V_1}\right)}\)
⇒ \(\frac{T_1-T_2}{T_1}\)
or \(\eta=1-\frac{Q_2}{Q_1}=1-\frac{T_2}{T_1}\)
Some Important Points Regarding Carnot Engine
The efficiency of an engine depends upon the temperatures between which it operates.
η is independent of the nature of the working substance.
η is one only if T2= 0. Since absolute zero is not attainable, hence even an ideal engine cannot be 100 % efficient.
η is one only if Q2= 0. But η = 1 is never possible even for an ideal engine. Hence Q2≠ 0. Thus it is impossible to extract heat from a single body and convert the whole of it into work.
If T2= T1, then η = 0
In actual heat engines, there are many losses due to friction, etc. and various processes during each cycle are not quasistatic, so the efficiency of actual engines is much less than that of an ideal engine.
Thermodynamics NEET Physics Class 11 Notes: Concepts and Formulas
Question 1. A Carnot engine has the same efficiency between
- 100 K and 500K and
- Tk and 900 K. The value of T is
Answer:
Efficiency η = \(1-\frac{T_2}{T_1}\)
or η = \(1-\frac{100}{500}\)
⇒ \(1-\frac{T}{900}\)
or \(\frac{100}{500}=\frac{T}{900}\)
∴ T = 180K
Question 2. A Carnot engine takes 103 kilocalories of heat from a reservoir at 627ºC and exhausts it to a sink at 27ºC. The efficiency of the engine will be.
Answer:
Efficiency of Carnot engine
η = \(1-\frac{T_2}{T_1}\)
⇒ \(1-\frac{300}{900}=\frac{2}{3}\)
or η = 66.6 %
Question 3. In the above problem, the work performed by the engine will be
Answer:
Work performed by the engine
W = \(\eta Q_1=\frac{2}{3} \times 10^6 \times 4.2\)
or W = 2.8 × 106 Joule
Question 4. A Carnot engine has an efficiency of 40% when the sink temperature is 27ºC. The source temperature will be
Answer:
⇒ \(\eta_{\text {efficiency }}=1-\frac{T_2}{T_1}\)
or \(\frac{2}{5}=1-\frac{300}{\mathrm{~T}_1}\)
∴ T1= 500K
Question 5. A reversible engine takes heat from a reservoir at 527ºC and gives out to the sink at 127ºC. The engine is required to perform useful mechanical work at the rate of 750 watts. The efficiency of the engine is
Answer:
Efficiency η = \(1-\frac{T_2}{T_1}\)
or η = \(1-\frac{400}{800}=\frac{1}{2}\)
η = 50%
Question 6. The efficiency of Carnot’s engine is 50%. The temperature of its sink is 7ºC. To increase its efficiency to 70%. The increase in heat of the source will be
Answer:
Efficiency in first state η = 50% = 1/2
T2= 273 + 7 = 280 K
Formula η = \(1-\frac{T_2}{T_1}\)
⇒ \(\frac{1}{2}=1-\frac{280}{\mathrm{~T}_1} \Leftrightarrow \frac{280}{\mathrm{~T}_1}=\frac{1}{2}\)
or T1 = 560ºK (temperature of source)
In the second state (1) \(\frac{70}{100}=1-\frac{280}{\mathrm{~T}_1}\)
∴ \(\mathrm{T}_1=\frac{2800}{3}=933.3 \mathrm{~K}\)
∴ Increase in source temperature = (933.3 – 560) = 373.3 K
Question 7. A Carnot’s engine works at 200ºC and 0ºC and another at 0ºC and –200ºC. The ratio of efficiency of the two is
Answer:
⇒ \(\eta=\frac{\left(T_1-T_2\right)}{T_1}\)
⇒ \(\eta_1=\frac{(473-273)}{473}=\frac{200}{473}\)
and \(\eta_2=\frac{(273-73)}{273}=\frac{200}{273}\)
⇒ \(\frac{\eta_1}{\eta_2}=\frac{273}{473}\)
= 0.577
Question 8. A Carnot engine works as a refrigerator in between 0ºC and 27ºC. How much energy is needed to freeze 10 kg of ice at 0ºC?
Answer:
Heat absorbed by the sink
Q2= 10 × 102 × 80 = 800 k.cal
Now \(\frac{Q_1}{Q_2}=\frac{T_1}{T_2}, Q_1=Q_2 \cdot \frac{T_1}{T_2}\)
∴ \(Q_1=800 \times \frac{300}{273} \mathrm{k} . \mathrm{cal}\)
= 879 kcal
Question 9. The work efficiency coefficient in the above question
Answer:
Work efficiency coefficient (coefficient of performance)
⇒ \(\beta=\frac{Q_2}{Q_1-Q_2}\)
⇒ \(\frac{800 \times 10^3}{(879-800) \times 10^3}\)
= 10.13
Question 10. A Carnot engine works as a refrigerator in between 250K and 300K. If it acquires 750 calories from a heat source at a low temperature, then the heat is generated at a higher temperature. (in calories) will be.
Solution:
⇒ \(\eta=\frac{Q_2}{Q_1-Q_2}=\frac{T_2}{T_1-T_2}\)
⇒ \(\frac{750}{Q_1-750}=\frac{250}{300-250}\)
Q1= 900 Calories
Question 11. A vessel of volume 2 x 10-2 m3 contains a mixture of hydrogen and helium at 47º C temperature and 4.15 x 105 N/m2 pressure. The mass of the mixture is 10-2 kg. Calculate the masses of hydrogen and helium in the given mixture.
Solution:
Let the mass of H2 is m1 and He is m2
∴ m1+ m2= 10-2 kg = 10 × 10-3 ….(1)
Let P1, P2 are partial pressure of H2 and He
P1+ P2= 4.15 × 105 N/m2
for the mixture
⇒ \(\left(P_1+P_2\right) V=\left(\frac{m_1}{n_1}+\frac{m_2}{n_2}\right) R T\)
⇒ \(4.15 \times 10^5 \times 2 \times 10^{-2}\)
⇒ \(\left(\frac{m_1}{2 \times 10^{-3}}+\frac{m_2}{4 \times 10^{-3}}\right) 8.31 \times 320\)
⇒ \(\frac{m_1}{2}+\frac{m_2}{4}=\frac{4.15 \times 2}{8.31 \times 320}=0.00312=3.12 \times 10^{-3}\)
⇒ 2m1+ m2= 12.48 × 10-3 kg …..(2)
Solving (1) and (2)
m1= 2.48 × 10-3 kg ≅2.5 × 10-3 kg
and m = 7.5 × 10-3 kg.
NEET Class 11 Physics Kinetic Theory of Gases Notes
Question 12. The pressure in a monoatomic gas increases linearly from 4 x 105 N m-2 to 8 x 105 N m-2 when its volume increases from 0.2 m3 to 0.5 m3. Calculate the following:
- Work done by the gas.
- Increase in the internal energy.
Answer:
1. As here pressure varies linearly with volume, work done by the gas
ΔW = ∫PdV = area under P-V curve
which in the light of Figure 1 becomes:
ΔW = PI(VF– VI) (PF– PI) × (VF– VI)
i.e., ΔW = \(P_I\left(V_F-V_I\right)+\frac{1}{2}\left(P_F-P_I\right) \times\left(V_F-V_I\right)\)

i.e., ΔW = \(\frac{1}{2}\)(0.5 – 0.2) (8 + 4) × 105
i.e., ΔW = 1.8 × 105 J
2. The change in internal energy of a gas is given by
ΔU = \(\mu C_V \Delta T=\frac{\mu R \Delta T}{(\gamma-1)}\)
⇒ \(\frac{\left(P_F V_F-P_I V_I\right)}{(\gamma-1)}\)
As the gas is monatomic γ = (5/3)
So, ΔU = \(\frac{10^5(8 \times 0.5-4 \times 0.2)}{[(5 / 3)-1]}\)
⇒ \(\frac{3}{2} \times 10^5(4-0.8)\)
i.e., ΔU = 4.8 × 105 J
Refrigerator Or Heat Pump
A refrigerator or heat pump is a heat engine that runs in revenue one direction.
It essentially consists of three parts
- Source: At higher temperature T1.
- Working substance: It is called refrigerant liquid ammonia and freon works as a working substance
- Sink: At lower temperature T2.

- The working substance takes heat Q2 from a sink (contents of the refrigerator) at a lower temperature, has a net amount of work done W in it by an external agent (usually the compressor of the refrigerator), and gives out a larger amount of heat Q1 to a hot body at temperature T1(usually atmosphere)
- Thus, it transfers heat from a cold to a hot body at the expense of mechanical energy supplied to it by an external agent. The cold is thus cooled more and more.
- The performance of a refrigerator is expressed using the “coefficient of performance” β which is defined as the ratio of the heat extracted from the cold body to the needed to transfer it to the hot body.
i.e \(\beta=\frac{\text { Heat extracted }}{\text { Work done }}\)
⇒ \(\frac{Q_2}{W}=\frac{Q_2}{Q_1-Q_2}\)
A perfect refrigerator transfers heat from a cold to a hot body without doing work e. W = 0 so that Q1=Q2 hence β = ∞
Carnot Refrigerator:
For Carnot refrigerator \(\frac{Q_1}{Q_2}=\frac{T_1}{T_2}\)
⇒ \(\frac{Q_1-Q_2}{Q_2}=\frac{T_1-T_2}{T_2}\)
or \(\frac{Q_2}{Q_1-Q_2}=\frac{T_2}{T_1-T_2}\)
So Coefficient of performance β = \(\frac{T_2}{\mathrm{~T}_1-\mathrm{T}_2}\)
⇒ \(\beta=\frac{T_2}{T_1-T_2}\)
here T1= temperature of surrounding T2 = temperature of cold body. It is clear that β = 0 when T2= 0 i.e. the coefficient of performance will be zero if the cold body is at a temperature equal to absolute zero.
Thermodynamics and Kinetic Theory of Gases NEET Physics Chapter 9 Notes
Relation between the coefficient of performance and efficiency of a refrigerator
We know β = \(\frac{Q_2}{Q_1-Q_2}=\frac{Q_2 / Q_1}{1-Q_2-Q_1}\)…………(1)
But the efficiency η = \(\frac{Q_2}{Q_1} \text { or } \frac{Q_2}{Q_1}=1-\eta\)…………(2)
Form (1) and (2) we get, \(\frac{1-\eta}{\eta}\)
Entropy
Entropy is a measure of the disorder of molecular motion of a system. The greater the disorder, the greater the entropy.
The change in entropy i.e
dS = \(=\frac{\text { Heat absorbed by system }}{\text { Absolute temperature }}\)
or \(\mathrm{dS}=\frac{\mathrm{dQ}}{\mathrm{T}}\)
The relation is called the mathematical form of the Second Law of Thermodynamics.
For solids and liquids
1. When heat is given to a substance changes its state at a constant temperature, then changes in entropy
⇒ \(d S=\frac{d Q}{T}= \pm \frac{m L}{T}\)
where the positive sign refers to heat absorption and the negative sign to heat evolution.
2. When heat given to a substance raises its temperature from T1 to T2 then changes in entropy
⇒ \(d S=\int \frac{d Q}{T}=\int_{T_1}^{T_2} m c \frac{d T}{T}=m c \log _e\left(\frac{T_2}{T_1}\right)\)
⇒ \(\Delta S=2.303 \mathrm{mc} \log _{10}\left(\frac{T_2}{T_1}\right)\)
For a perfect gas: The perfect gas equation for n moles is PV = nRT
ΔS = \(\int \frac{d Q}{T}=\int \frac{\mu C_V d T+P d V}{T}\) [As dQ = dU+dW]
ΔS = \(\int \frac{\mu C_V d T+\frac{\mu R T}{V} d V}{T}\)
⇒ \(\mu C_V \int_{T_1}^{T_2} \frac{d T}{T}+\mu R \int_{V_1}^{V_2} \frac{d V}{V}\) [ As PV = μ RT]
∴ ΔS = \(\mu C_V \text { long }_e\left(\frac{T_2}{T_1}\right)+\mu \log _e\left(\frac{V_2}{V_1}\right)\)
In terms of T and P, ΔS = \(m C_p{lon}_g\left(\frac{T_2}{T_1}\right)-\mu R \log _e\left(\frac{P_2}{P_1}\right)\)
and in terms of P and V ΔS = \(\mu C_V \log _e\left(\frac{P_2}{P_1}\right)+\mu C_p \log _e\left(\frac{V_2}{V_1}\right)\)