Chemical Properties Of Group-13 Elements
The members of group-13 elements have three electrons in their valence shells. Except for the last member, Tl, all other members use these electrons to form three bonds and thus exhibit an oxidation state of +3. In +3 oxidation state.
The Members Of The Boron Family Are Expected To Form Covalent Bonds For The Following Reasons:
- Small size and high charge (+3) cause high polarisation of the anions, leading to the formation of covalent bonds.
- The large value of the sum of the first three ionisation enthalpies, ΔiH1< ΔiH2 < ΔiH3 of these elements also suggests that the bonds will be largely covalent.
- The difference in electronegativity between the elements of group 13 and those of the higher groups is not very significant. This fact also agrees with the formation of covalent bonds.
Because of its small size and high ionisation enthalpies, it is not possible for boron to form B3+ ions by losing its three valence electrons. Therefore, Boron does not form ionic compounds. In fact, it always forms covalent compounds by sharing its valence electrons. The sum of the first three ionisation enthalpies, ΔiH1< ΔiH2 < ΔiH3 of Al is also higher
Read and Learn More CBSE Class 11 Chemistry Notes
But less than that of B. So Al also has a strong tendency to form covalent compounds,
Chemical Properties of Group-13 Elements Class 11 Chemistry
The Members Of The Boron Family Are Expected To Form Covalent Bonds For example:
AlCl3, AlBrv and AlI3. Like Al, compounds of the rest of the members such as GaCl3, InCl3 etc. are covalent when anhydrous. However, all the members except B form metal ions in solution.
This change from covalent to ionic nature may be explained by the fact that in aqueous solutions these ions undergo hydration and the amount of hydration enthalpy exceeds the ionisation enthalpy. Ga, In and Tl show two oxidation states of +1 and +3 due to the inert pair effect. The compounds in the +1 oxidation state are more ionic than the compounds in the +3 oxidation state.
In a trivalent state, the number of electrons in the valence shell of the central atom in a molecule of these elements is only six (two electrons less than the octet) and therefore, such electron-deficient molecules behave as Lewis acids. For example, BCl3 (Lewis acid) readily accepts an unshared pair of electrons from ammonia (Lewis base) to form the adduct, BCl3 -NH3
1. Reaction With Dioxygen Or Air
1. All the members of group 13 react with dioxygen at higher temperatures to form trioxides of the general formula M2O3. Tl forms both T1203 and some amount of Tl3O
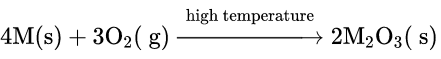
The reaction of Al with O2 is known as a thermite reaction which is highly exothermic (ΔH0 =-1670kJ mol-1 ). A very strong affinity of Al for oxygen is used in the extraction of other metals from their oxides (thermite process). For example, Mn and Cr can be extracted from Mn304 and Cr2O3 respectively by this process.
Group 13 Elements Chemical Properties Class 11 Notes
2. The reactivity of group-13 elements towards dioxygen increases on moving down the group. Pure crystalline boron is almost unreactive towards air at ordinary temperature. Al does not react with dry air. However, it gets tarnished readily in moist air even at ordinary temperatures due to the formation of a thin oxide (Al2O3) layer on the surface which prevents the metal from further reaction. When amorphous boron and aluminium metal are heated in air, they form boron trioxide and aluminium trioxide (Al2O3) respectively.
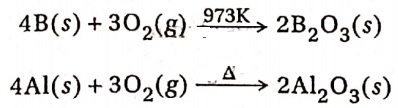
Ga and In are not affected by air but Tl forms an oxide on its surface in the presence of air
3. B and Al react with dinitrogen at high temperatures to form the corresponding nitrides
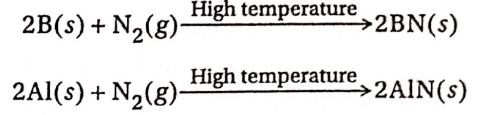
Ga, In and Tl do not react with N2 to form the corresponding nitrides.
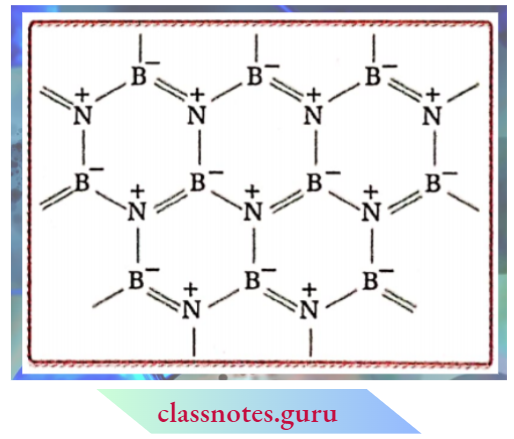
4. Boron nitride is a white slippery solid which melts under pressure at 3246K. It is chemically inert towards the air, oxygen, hydrogen, chlorine, etc. even on heating. The total number of valence electrons of one B and one N- atom is equal to the number of valence electrons of two C-atoms.
5. Therefore, the structure of boron nitride is almost the same as that of graphite having a layer lattice. In each layer, alternate B and N-atoms (both sp² -hybridised) form a planar hexagon The layers are stacked over one another in such a way that the N-atom of one layer is directed over the B-atom of another layer. Because of its structural similarity with graphite, boron nitride is also called inorganic graphite
Chemical Properties of Boron and Other Group 13 Elements
When boron nitride is heated at 1800°C under very high pressure, it gets converted to a cubic form comparable to diamond. This extremely hard variety known as borazon is used for cutting diamonds.
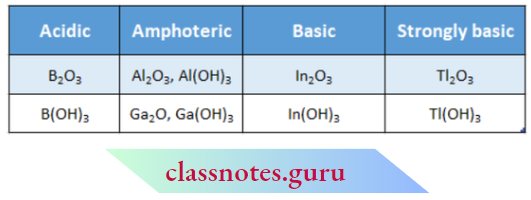
The Acid-Base Character Of Oxides And Hydroxides
1. Trioxides of the elements of the boron family react with water to form their corresponding hydroxides.
M2O3 + 3H2O → 2M(OH)3
2. The nature of these oxides and hydroxides changes on moving down the group. Both B203 and B(OH)3 are weakly acidic. They dissolve in alkali to form metal borates.
B2O3, + 2NadH → 2NaBO2(sodium metaborate) +H2O
B(OH)3+ 3NaOH → Na3BO3(sodium borate) + 3H2O
Aluminium oxide and hydroxide are amphotericin in nature. Both of them dissolve in alkalies as well as acids.
Al2O3(s) + 3H2SO4(aq)→ Al2(SO4)3(aq) + 3H2O(l)
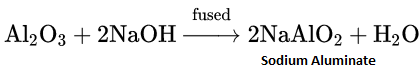
Similarly, Al(OH)3(s) + NaOH(s)→Na[Al(OH)4](aq)
Al(OH)3(aq) + 3HCl(aq)→AlCl3 (aq) + 3H2O(l)
The oxide and hydroxide of Ga are also amphoteric while those of Tl are basic.
Therefore, the basic character of oxides and hydroxides increases down the group
NCERT Class 11 Chemistry Group 13 Elements Chemical Properties
3. Thallium Forms Two Types Of Hydroxides:
Thallic hydroxide [Tl(OH)3] and thallous hydroxide (TlOH). Tl(OH)3 is insoluble in H2O but TlOH is soluble and is a strong base like alkali metalhydroxides.
Hydroxides Explanation:
On moving down the group, the magnitude of ionisation enthalpy decreases. As a result, the strength of the M—O bond also decreases and therefore, its cleavage becomes progressively easier resulting in the increased basic strength down the group.
An extremely hard crystalline form of aluminium oxide called corundum is used as an abrasive.It can be made by heating amorphous aluminium oxide at about 2000K. Aluminium forms a series of mixed oxides with other j metals, some of them occurring naturally as semi-precious stones. These include ruby (Cr2+) and blue sapphire (CO2+, Fe2+, Tl4+)
2. Reaction With Hydrogen
1. Group-13 elements form hydrides ofthe type MH3. The members of the boron family do not combine directly with hydrogen. However, several hydrides are known which can be prepared indirectly. Boron forms several stable covalent hydrides which are collectively called boranes.
The Two Most Important Types Of Boranes Are As Follows:
The members of the boron family do not combine directly with hydrogen. However, several hydrides are known which can be prepared indirectly. Boron forms several stable covalent hydrides, which are collectively called boranes.
The Two Most Important Types Of Boranes Are As Follows:
1. Boranes with general formula BnHn+4 are called nido-boranes.
Types Of Boranes Example: Diborane (B2H6) pentaborane-9 (B5H9).
Boranes with the general formula BnHn+6 are called arachno boranes
Types Of Boranes Example: Tetraborane (B4H10),pentaborane-11 (B5H11).
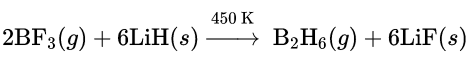
2. The simplest hydride is diborane (B2H6) which is prepared by the reaction of BF3 with lithium hydride industrially.
Chemical Properties of Group-13 Elements NCERT Solutions Class 11
3. The other members of group 13 also form several hydrides which are polymeric.
For example: (AlH3)R, (GaH3)n, (InH3)n.
The stability of these hydrides decreases down the group and thallium hydride is quite unstable.
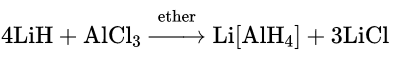
Boron, aluminium and gallium also form complex anionic hydrides such as NaBH4 (sodium borohydride), LiAlH4 (lithium aluminium hydride) and LiGaH4 (lithium gallium hydride). These complex hydrides act as powerful reducing agents.
4. The hydrides are weak Lewis acids and readily form adducts with strong Lewis bases to form compounds of the type MH3:B (B = base).
For example: AlH4 :NMe3, GaH4
NMe etc. NMe3 + AlH3→ [Me4N: → AlH3]
3. Reaction With Acids And Alkalies
The Action Of Acids:
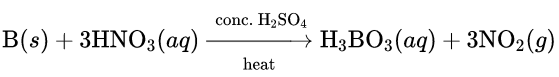
1. Boron remains inert in the presence of non-oxidising acids such as HCl. However, it undergoes oxidation by strong oxidising acids such as a mixture of hot concentrated H3SO4 and HNO3 (2: 1) to form boric acid (H3BO3) at very high temperature
Group 13 Elements Reaction and Properties Class 11 Chemistry
2. The remaining elements of this group react with both oxidising and non-oxidising acids. For example, Al dissolves in dilute HCl and liberates dihydrogen
2Al(s) + 6HCl(aq) → 2Al2+(aq) + 6Cl–(aq) + 3H2(g)
3. Concentrated nitric acid renders aluminium passive by forming a protective layer of its oxide (Al2O33) on the surface of the metal. Thus aluminium vessels can be used to store concentrated HNO3
2A1 + 6HNO3→ Al2O3 + 6NO2 + 2H2>O
Ga, In and TI react with dilute acids to liberate H3
Action Of Alkalies:
1. When boron is fused with alkalies (NaOH or KOH) at a temperature greater than 775K, it forms borates and liberates dihydrogen
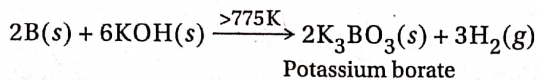
2. Boron dissolves in a fused mixture of Na2CO3 and NaNO3 at 1123K to produce borate and nitrite salt and liberates carbon dioxide
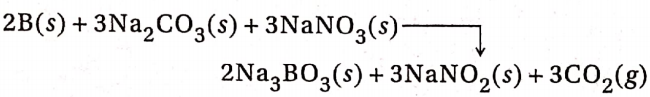
3. Al and Ga also react with aqueous alkalies with the evolution of dihydrogen.
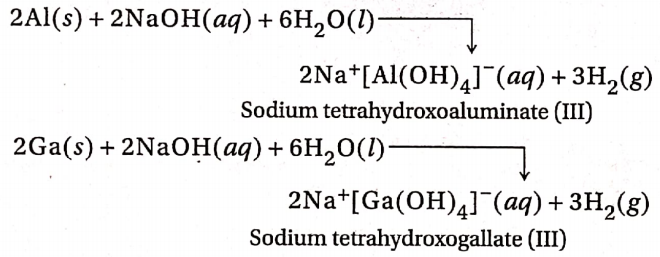
In and Tl do not react with alkalis
CBSE Class 11 Chemistry Group-13 Elements Chemical Reactions
4. Reaction With Halogens
Elements of group-13 react with halogens at high temperatures to produce trihalides of the general formula, MX3. However, thallium (III) iodide doesn’t exist
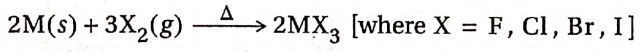
Trihalides Of Boron:
Due to its small atomic size and high, ionisation enthalpy, boron forms covalent trihalides
BX3. BF3 is a gas, BCl3 and BBr3 are liquids and BI3 is a solid. All these are trigonal planar molecules in which the central B -atom is sp² -hybridised. The three unpaired electrons of p -orbitals of three halogen atoms overlap with the three sp2 -orbitals of boron to form three sp²-p, B—X, σ -bonds. The unhybridised empty p-orbital remains perpendicular to the plane of the molecules
Since there are only six electrons in the valence shell ofthe central boron atom in boron trihalides, they can accept two more electrons to acquire a stable octet, i.e., boron trihalides can behave as Lewis acids. The Lewis acid character, however, decreases in the order:
BI > BBr3 > BCl3 > BF3.
Trihalides Of Boron Explanation:
This order of relative Lewis acid strength of boron trihalides, which is just the reverse of what may be expected based on the electronegativities of the halogen atoms, can well be explained based on the tendency of the halogen atom to donate its lone pair of electrons to the boron atom through pn-pn back bonding.
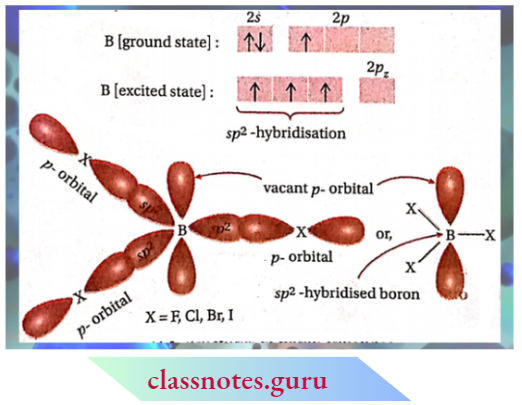
Since the vacant 2p -orbital of B and the 2p-orbital of Fatom containing a lone pair of electrons are equal in size, therefore, the tendency of the F -atom to donate the unshared pair by pn-pn back bonding is maximum.
BF3 can well be represented as a resonance hybrid of four resonating structures As a result of resonance involving pn-pn back bonding, the electron density on the boron atom increases effectively and so its strength as a Lewis acid decreases considerably.
Chemical Behavior of Group 13 Elements Class 11
As the size of the halogen atom increases on going from Cl to I, the extent of overlap between the 2p-orbital of boron and a large p-orbital of halogen (3p of Cl, 4p of Br and 5p of I] decreases. As a consequence, the electron deficiency strength decreases on going from BF3 to BI3
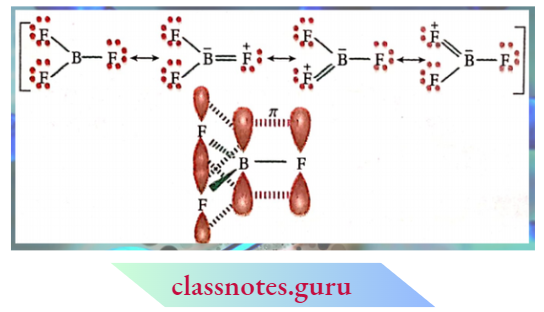
Halides Of Aluminium:
The halides of aluminium in the vapour state, as well as in an inert solvent such as benzene, exist ns dimers. For example, AlCl3 exists as Al2Cl6.
Halides Of Aluminium Explanation:
In AlCl3, there are six electrons (two electrons less than the tyre octet) around the central Al-atom. In the dimeric structure, each Al completes its octet by accepting a lone pair of electrons from the Cl-atom of another AlCl3 molecule. The dimeric form exists lit vapour state at < 473K. However, at higher temperatures, it dissociates to a trigonal planar AlCl3 molecule.
In polar solvents such as water, the dimer dissociates and it is the high hydration enthalpy which helps this dissociation leading to the formation of Al3+ion.
Class 11 Chemistry Group 13 Elements Chemical Reactions and Properties
Al3Clg + 6H2O ⇌ 2[Al(H2O)6]3++(aq) + 6Cl–(aq)
Therefore, anhydrous AlCl3 is covalent but, hydrated aluminium chloride is ionic.
Unlike aluminium halides, boron halides exist as monomers and this is because the boron atom is so small that cannot accommodate four large-sized halogen atoms
around it.
